Przejdź do
Wybierz wielkość
| Do Państwa/SKU | Dostępność | Cena netto |
|---|---|---|
50 mg | Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności | 1000,00 zł |
Informacje o tej pozycji
1000,00 zł
Quality Segment
form
solid
solubility
DMSO: 224 mg/mL at 25 °C
antibiotic activity spectrum
Gram-positive bacteria
mode of action
protein synthesis | interferes
storage temp.
2-8°C
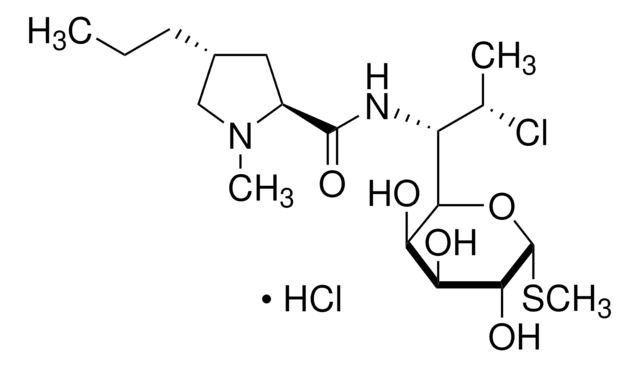
SMILES string
CCC[C@@H]1C[C@H](N(C)C1)C(=O)NC([C@H](C)Cl)C2O[C@H](SC)[C@H](OP(O)(O)=O)[C@@H](O)[C@H]2O
InChI
1S/C18H34ClN2O8PS/c1-5-6-10-7-11(21(3)8-10)17(24)20-12(9(2)19)15-13(22)14(23)16(18(28-15)31-4)29-30(25,26)27/h9-16,18,22-23H,5-8H2,1-4H3,(H,20,24)(H2,25,26,27)/t9-,10+,11-,12?,13+,14-,15?,16+,18+/m0/s1
InChI key
UFUVLHLTWXBHGZ-MWBQRTRKSA-N
General description
Application
Biochem/physiol Actions
Mode of Action: Inhibits protein synthesis in bacterial by binding the 50s ribosomal subunit.
Other Notes
1 of 1
Ta pozycja | |||
|---|---|---|---|
| mode of action protein synthesis | interferes | mode of action protein synthesis | interferes | mode of action protein synthesis | interferes | mode of action protein synthesis | interferes |
| antibiotic activity spectrum Gram-positive bacteria | antibiotic activity spectrum Gram-negative bacteria, Gram-positive bacteria | antibiotic activity spectrum Gram-positive bacteria | antibiotic activity spectrum Gram-positive bacteria |
| Quality Level 100 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| form solid | form powder or crystals | form - | form powder or crystals |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| solubility DMSO: 224 mg/mL at 25 °C | solubility H2O: 50 mg/mL | solubility H2O: soluble 50 mg/mL | solubility H2O: 50 mg/mL, clear, colorless to faintly yellow |
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Lact. - Skin Sens. 1
Klasa składowania
11 - Combustible Solids
wgk
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.