おすすめの製品
product name
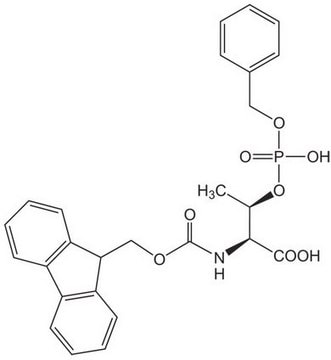
Fmoc-Ser(PO(OBzl)OH)-OH, Novabiochem®
品質水準
製品種目
Novabiochem®
アッセイ
≥90.0% (acidimetric)
≥95.0% (HPLC)
≥97% (TLC)
形状
powder
反応適合性
reaction type: Fmoc solid-phase peptide synthesis
メーカー/製品名
Novabiochem®
アプリケーション
peptide synthesis
官能基
Fmoc
保管温度
−20°C (−15°C to −25°C)
InChI
1S/C25H24NO8P/c27-24(28)23(16-34-35(30,31)33-14-17-8-2-1-3-9-17)26-25(29)32-15-22-20-12-6-4-10-18(20)19-11-5-7-13-21(19)22/h1-13,22-23H,14-16H2,(H,26,29)(H,27,28)(H,30,31)/t23-/m0/s1
InChI Key
ZBPUWGDUVAAWJY-QHCPKHFHSA-N
関連するカテゴリー
詳細
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] T. Wakamiya, et al. (1994) Chem. Lett., 1099.
[2] P. White & J. Beythien in ′Innovations & Perspectives in Solid Phase Synthesis and Combinatorial Libraries, 4th International Symposium′, Mayflower Scientific Ltd., Birmingham, 1996, pp. 557.
[3] H. Schmid, et al., Poster 423 presented at the 15th American Peptide Symposium, Nashville, 1997.
[4] T. L. Gururaja & M. J. Levine (1996) Pept. Res., 9, 283.
[5] T. Vorherr, et al. (1995) Bioorg. Med. Chem. Lett., 5, 2661.
[6] G. Shapiro, et al. (1996) Bioorg. Med. Chem. Lett., 6, 409.
[7] M. John, et al. (1996) Pept. Res., 9, 71.
[8] K. Teruya, et al. (2004) J. Pept. Sci., 10, 479.
[9] T. J. Attard, et al. (2009) Int. J. Pept. Res. Ther., 15, 69.
関連事項
アナリシスノート
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.0 % (a/a)
Purity (TLC(CMA1)): ≥ 97 %
Assay (HPLC, area%): ≥ 95.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Assay (acidimetric): ≥ 90.0 %
Water (K. F.): ≤ 1.0 %
Ethyl acetate (HS-GC): ≤ 0.5 %
Acetate (IC): ≤ 0.1 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
法的情報
関連製品
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
この製品を見ている人はこちらもチェック
プロトコル
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)