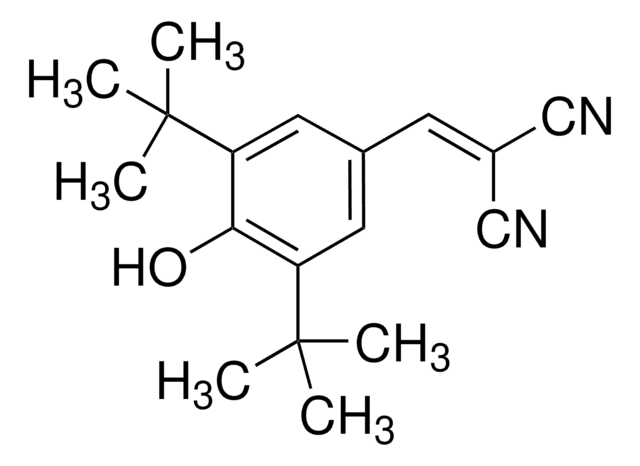
5.05602
BDK Inhibitor II, (S)-CPP
別名:
BDK Inhibitor II, (S)-CPP, (S)-α-Chloro-phenylpropionic acid, BCKD Kinase Inhibitor II, Branched-Chain α-Ketoacid Dehydrogenase Kinase Inhibitor II, BCKD Kinase Inhibitor II, Branched-Chain α-Ketoacid Dehydrogenase Kinase Inhibitor II, (S)-α-Chloro-phenylpropionic acid
ログイン組織・契約価格を表示する
すべての画像(1)
About This Item
おすすめの製品
アッセイ
≥98% (HPLC)
品質水準
形状
oil
メーカー/製品名
Calbiochem®
保管条件
OK to freeze
desiccated
protect from light
色
clear colorless
溶解性
DMSO: 100 mg/mL
保管温度
−20°C
詳細
A cell-permeable chlorophenylpropionate compound that is superior to its structural analog 4PB (Cat. No. 567616) as an inhibitor against mitochondrial branched-chain α-ketoacid dehydrogenase complex (BCKDC) regulatory kinase BDK (BCKD kinase; IC50 = 6.3 vs. 53.1 M) due to much optimized affinity toward the BDK N-terminal allosteric site (Kd = 2.4 vs. 5.7 M), effectively blocking BDK substrate access by preventing BDK interaction with the homo-24-meric dihydrolipoyltransferase BCKDC E2 component, while exhibiting little potency against PDK2 or BCKD phosphatase BDP even at concentrations as high as 1 mM (<10% inhibition). Shown to effectively reduce E1-α Ser293 phosphorylation with concomitant BCKDC activity upregulation in multiple tissues in mice (EDmax =160 mg/kg via i.p.) in vivo. Despite good stability and low in vivo clearance , high doses are recommended for mice treatment due to low tissue penetration efficiency.
A cell-permeable chlorophenylpropionate compound that is superior to its structural analog 4PB (Cat. No. 567616) as an inhibitor against mitochondrial BCKDC (branched-chain α-ketoacid dehydrogenase complex) regulatory kinase BDK (BCKD kinase; IC50 = 6.3 vs. 53.1 µM) due to much optimized affinity toward the BDK N-terminal allosteric site (Kd = 2.4 vs. 5.7 µM) via carboxylate oxygens-mediated hydrogen bonds, as well as hydrophobic interactions mediated by the α-chlorine and the phenyl ring, effectively blocking BDK substrate access by preventing BDK interaction with the homo-24-meric dihydrolipoyltransferase BCKDC E2 component, while exhibiting little potency against PDK2 (pyruvate dehydrogenase kinase isoform 2) or BDP (BCKD phosphatase) even at concentrations as high as 1 mM (<10% inhibition). Shown to effectively reduce E1-α Ser293 phosphorylation with concomitant BCKDC activity upregulation in multiple tissues in mice (EDmax =160 mg/kg via i.p.; 60 min) in vivo, resulting in significant downregulation of serum BCAA (branched-chain amino acid) Leu/Ile and Val levels. Pharmacokinetic studies reveal good stability in vitro (half-life = 3.1 h and 6.8 h using murine liver S9 preparation or murine hepatocytes, respectively) and low clearance in vivo (CL/F = 0.113 mL/min; 40 mg/kg i.p.), however the compound does not penetrate tissues efficiently (VZ/F = 20.8 mL; 40 mg/kg i.p.) and high dosages are recommended for mice treatment.
生物化学的/生理学的作用
Cell permeable: yes
Primary Target
BDK
BDK
Reversible: yes
包装
Packaged under inert gas
警告
Toxicity: Standard Handling (A)
再構成
Following reconstitution, aliquot and freeze (-20°C). Stock solutions are stable for up to 1 month at -20°C.
Use only fresh DMSO for reconstitution.
その他情報
Tso, S.C., et al. 2013. Proc. Natl. Acad. Sci. USA110, 9728.
法的情報
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)



![6-[4-(2-ピペリジン-1-イルエトキシ)フェニル]-3-ピリジン-4-イルピラゾロ[1,5-a]ピリミジン ≥98% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/337/374/32fd5b47-5df0-4380-8347-33475bc58d2f/640/32fd5b47-5df0-4380-8347-33475bc58d2f.png)