PHR1747
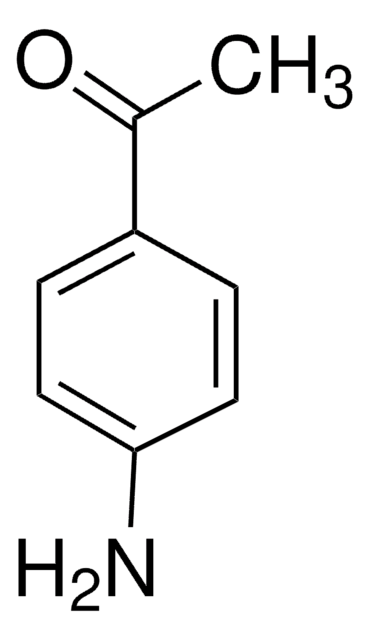
4′-Aminoacetophenone
Pharmaceutical Secondary Standard; Certified Reference Material
Sinonimo/i:
4′-Aminoacetophenone, 4-Acetylaniline
About This Item
Prodotti consigliati
Grado
certified reference material
pharmaceutical secondary standard
Livello qualitativo
Famiglia di API
clenbuterol
CdA
current certificate can be downloaded
Confezionamento
pkg of 200 mg
tecniche
HPLC: suitable
gas chromatography (GC): suitable
P. eboll.
293 °C (lit.)
Punto di fusione
103-107 °C (lit.)
applicazioni
pharmaceutical (small molecule)
Formato
neat
Temperatura di conservazione
2-30°C
Stringa SMILE
CC(=O)c1ccc(N)cc1
InChI
1S/C8H9NO/c1-6(10)7-2-4-8(9)5-3-7/h2-5H,9H2,1H3
GPRYKVSEZCQIHD-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Applicazioni
Risultati analitici
Altre note
Nota a piè di pagina
Prodotti correlati
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Choose from one of the most recent versions:
Certificati d'analisi (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.