213136
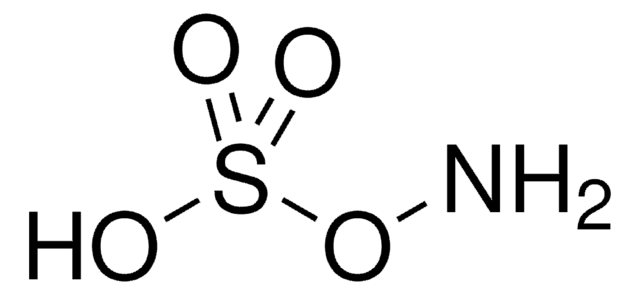
Hydroxylamine-O-sulfonic acid
97%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
H2NOSO3H
Numero CAS:
Peso molecolare:
113.09
Numero CE:
Numero MDL:
Codice UNSPSC:
12352300
ID PubChem:
NACRES:
NA.21
Prodotti consigliati
Livello qualitativo
Saggio
97%
Punto di fusione
210 °C (dec.) (lit.)
Temperatura di conservazione
2-8°C
Stringa SMILE
NOS(O)(=O)=O
InChI
1S/H3NO4S/c1-5-6(2,3)4/h1H2,(H,2,3,4)
DQPBABKTKYNPMH-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
Hydroxylamine-O-sulfonic acid is a versatile synthetic reagent. It is widely employed as a nucleophile and as an electrophile in various organic syntheses. It can be synthesized by reacting hydroxylamine sulfate with 30% fuming H2SO4.
Applicazioni
Hydroxylamine-O-sulfonic acid (HOSA) may be employed in the preparation of the following:
- monosubstituted and 1,1-disubstituted hydrazines
- symmetrically substituted pyrroles
- aniline
- C-substituted amino derivatives
- lactams
Hydroxylamine-O-sulfonic acid may be used in the following processes:
As a nitrogen transfer reagent for the conversion of aldehydes to nitriles in aqueous medium.
Synthesis of N-aminopiperidine (NAPP) by reacting with piperidine.
As a nitrogen transfer reagent for the conversion of aldehydes to nitriles in aqueous medium.
Synthesis of N-aminopiperidine (NAPP) by reacting with piperidine.
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Direct synthesis of nitriles from aldehydes with hydroxylamine-O-sulfonic acid in acidic water.
Quinn DJ, et al.
Tetrahedron Letters, 57(34), 3844-3847 (2016)
A New Strategy for the Preparation of N-Aminopiperidine Using Hydroxylamine-O-Sulfonic Acid: Synthesis, Kinetic Modelling, Phase Equilibria, Extraction and Processes."
Labarthe E, et al.
Advances in Chemical Engineering and Science, 3(2) (2013)
Hydroxylamine-O-sulfonic Acid.
Erdik E and Saczewski J.
e-EROS Encyclopedia of Reagents for Organic Synthesis. (2013)
Synthetic Methods and Reactions; 671. One-Step Conversion of Alicyclic Ketones into Lactams with Hydroxylamine-O-sulfonic Acid/Formic Acid.
Olah GA and Fung AP.
Synthesis, 07, 537-538 (1979)
K Sakano et al.
Mutation research, 479(1-2), 101-111 (2001-07-27)
2-Nitropropane (2-NP), a widely used industrial solvent, is carcinogenic to rats. To clarify the mechanism of carcinogenesis by 2-NP, we investigated DNA damage by 2-NP metabolites, N-isopropylhydroxylamine (IPHA) and hydroxylamine-O-sulfonic acid (HAS), using 32P-5'-end-labelled DNA fragments obtained from genes that
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.