F6129
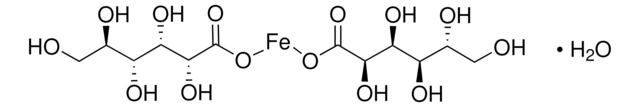
Iron(III) citrate
technical grade
Sinonimo/i:
Ferric citrate, Iron(III) citrate, Iron(III) citrate hydrate
Scegli un formato
Scegli un formato
About This Item
Prodotti consigliati
Grado
technical grade
Livello qualitativo
Stato
powder
Composizione
Fe, 16.5-18.5%
tecniche
cell culture | mammalian: suitable
applicazioni
battery manufacturing
Stringa SMILE
OC12CC(=O)O[Fe](OC(=O)C1)OC2=O
InChI
1S/C6H8O7.Fe/c7-3(8)1-6(13,5(11)12)2-4(9)10;/h13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);/q;+3/p-3
NPFOYSMITVOQOS-UHFFFAOYSA-K
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
It can also be used in the degradation of tetracycline for the treatment of polluted water.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 2
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Plasmonic nanoparticles have unique optical properties that can be tailored to suit a variety of applications in the biotechnology1–8 and electronics9–16 industries.
Filtri attivi
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.