218693
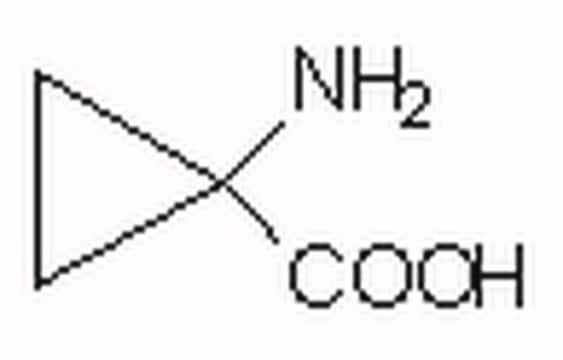
1-Aminocyclohexanecarboxylic acid
98%
Sinonimo/i:
Homocycloleucine
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
H2NC6H10CO2H
Numero CAS:
Peso molecolare:
143.18
Beilstein:
2355692
Numero CE:
Numero MDL:
Codice UNSPSC:
12352106
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
98%
Forma fisica
solid
Impiego in reazioni chimiche
reaction type: solution phase peptide synthesis
Punto di fusione
>300 °C (lit.)
applicazioni
peptide synthesis
Stringa SMILE
NC1(CCCCC1)C(O)=O
InChI
1S/C7H13NO2/c8-7(6(9)10)4-2-1-3-5-7/h1-5,8H2,(H,9,10)
WOXWUZCRWJWTRT-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
A Romanelli et al.
Journal of peptide science : an official publication of the European Peptide Society, 7(1), 15-26 (2001-03-14)
Secondary structure formation and stability are essential features in the knowledge of complex folding topology of biomolecules. To better understand the relationships between preferred conformations and functional properties of beta-homo-amino acids, the synthesis and conformational characterization by X-ray diffraction analysis
Wioleta Kowalczyk et al.
Journal of medicinal chemistry, 47(24), 6020-6024 (2004-11-13)
The synthesis and some pharmacological properties of two sets of analogues, one consisting of six peptides with 1-aminocyclohexane-1-carboxylic acid (Acc) in position 2 and the other with the amino acid in position 3, have been described. All the peptides were
Fernando Formaggio et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 8(1), 84-93 (2002-02-02)
The achiral, nitroxyl-containing alpha-amino acid TOAC (TOAC = 2,2,6,6-tetramethylpiperidine-1-oxyl-4-amino-4-carboxylic acid), in combination with the chiral alpha-amino acid C(alpha)-methyl valine [(alphaMe)Val], was used to prepare short peptides (from di- to hexa-) that induced the enantioselective oxidation of racemic 1-phenylethanol to acetophenone.
Mitsunobu Doi et al.
Biochemical and biophysical research communications, 297(1), 138-142 (2002-09-11)
Endomorphin (EM2, Tyr-Pro-Phe-Phe-NH(2)) can assume various conformations related to cis/trans-rotamers of the amide linkage of Tyr-Pro. To control isomerization, restricted or flexible components have been introduced at the Pro position. We focused on [Chx(2)]EM2, an EM2 analogue substituting 1-aminocyclohexane-1-carboxlylic acid
Olga Labudda-Dawidowska et al.
Journal of medicinal chemistry, 48(25), 8055-8059 (2005-12-13)
In the present work, a sterically constrained noncoded amino acid, 1-aminocyclohexane-1-carboxylic acid (Acc), was substituted in position 8 of the peptide chain of bradykinin (BK) and position 6, 7, or 8 of its B2 receptor antagonist [D-Arg0,Hyp3,Thi,(5,8)D-Phe7]BK, previously synthesized by
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.