推荐产品
等級
certified reference material
pharmaceutical secondary standard
品質等級
agency
traceable to BP 741
traceable to Ph. Eur. E0109000
traceable to USP 1235300
API 家族
enalapril
CofA
current certificate can be downloaded
技術
HPLC: suitable
gas chromatography (GC): suitable
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-30°C
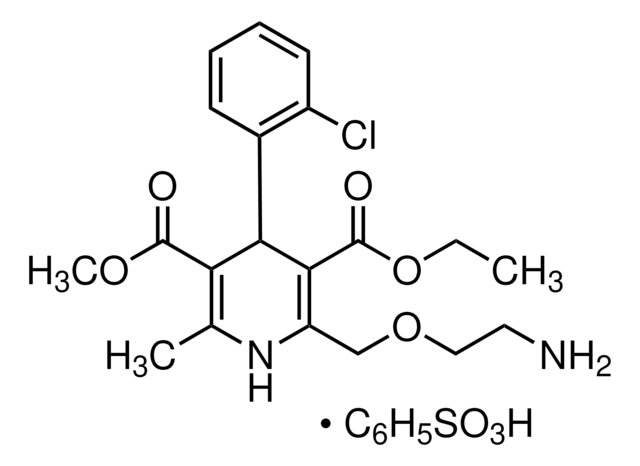
SMILES 字串
C[C@@H](C(N1[C@H](C(O)=O)CCC1)=O)N[C@H](CC(OCC)=O)CCC2=CC=CC=C2.O=C(OO)/C=C\C(O)=O
InChI
1S/C21H30N2O5.C4H4O5/c1-3-28-19(24)14-17(12-11-16-8-5-4-6-9-16)22-15(2)20(25)23-13-7-10-18(23)21(26)27;5-3(6)1-2-4(7)9-8/h4-6,8-9,15,17-18,22H,3,7,10-14H2,1-2H3,(H,26,27);1-2,8H,(H,5,6)/b;2-1-/t15-,17-,18-;/m0./s1
InChI 密鑰
UQKQUZOIQFOJOI-KJHLASMFSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards. Enalapril Maleate is a drug, which is widely used as an antihypertensive and a vasodilator in congestive heart failure. It is a derivative of 2 amino acids such as L-alanine and L-proline.
應用
Enalapril Maleate may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using derivative UV spectrophotometry and chromatography techniques.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
生化/生理作用
一种长效血管紧张素转换酶抑制剂。
分析報告
These secondary standards offer multi-traceability to the USP, EP (PhEur) and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
腳註
To see an example of a Certificate of Analysis for this material enter LRAC3898 in the slot below. This is an example certificate only and may not be the lot that you receive.
相關產品
产品编号
说明
价格
訊號詞
Warning
危險聲明
危險分類
Repr. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 2
其他客户在看
Determination of hydrochlorothiazide and enalapril maleate in tablet formulations by reversed-phase HPLC
Al-Momani GI
Turkish Journal of Chemistry, 25(1), 49-54 (2001)
Simultaneous determination of enalapril maleate and hydrochlorothiazide in tablets by derivative UV spectrophotometry and high-performance liquid chromatography
Carlucci G, et al.
International Journal of Pharmaceutics, 93(1-3), 245-248 (1993)
Fernando Arturo Reyes-Marín et al.
Revista de investigacion clinica; organo del Hospital de Enfermedades de la Nutricion, 64(4), 315-321 (2012-12-12)
Residual renal function (RRF) is an important determinant of mortality and morbidity in patients receiving peritoneal dialysis (PD). Recent studies have shown a positive effect of angiotensin converting enzyme inhibitors (ACEi) and angiotensin II receptor blockers (ARBs) on RRF in
Justin Brewer et al.
Hypertension (Dallas, Tex. : 1979), 62(5), 886-892 (2013-09-18)
Hypertension during preeclampsia is associated with increased maternal vascular sensitivity to angiotensin II (ANGII). This study was designed to determine mechanisms whereby agonistic autoantibodies to the ANGII type I receptor (AT1-AA) enhance blood pressure (mean arterial pressure [MAP]) and renal
Donald E Stull et al.
Value in health : the journal of the International Society for Pharmacoeconomics and Outcomes Research, 16(1), 164-176 (2013-01-23)
To present a step-by-step example of the examination of heterogeneity within clinical trial data by using a growth mixture modeling (GMM) approach. Secondary data from a longitudinal double-blind clinical drug study were used. Patients received enalapril or placebo and were
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门