所有图片(1)
About This Item
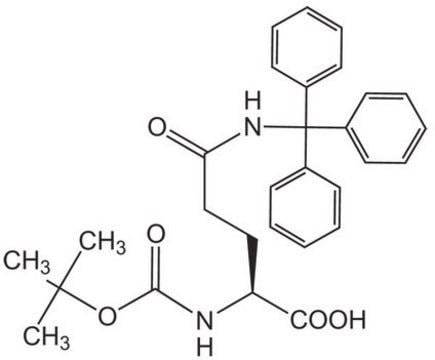
经验公式(希尔记法):
C39H34N2O5
CAS号:
分子量:
610.70
Beilstein:
4343953
MDL號碼:
分類程式碼代碼:
12352209
eCl@ss:
32160406
PubChem物質ID:
NACRES:
NA.26
推荐产品
产品名称
FFmoc-N-三苯甲基-L-谷氨酰胺, ≥98.0% (HPLC)
品質等級
化驗
≥98.0% (HPLC)
形狀
powder
光學活性
[α]/D -14.0±1.5°, c = 1% in DMF
反應適用性
reaction type: Fmoc solid-phase peptide synthesis
應用
peptide synthesis
官能基
Fmoc
儲存溫度
2-8°C
SMILES 字串
OC(=O)[C@H](CCC(=O)NC(c1ccccc1)(c2ccccc2)c3ccccc3)NC(=O)OCC4c5ccccc5-c6ccccc46
InChI
1S/C39H34N2O5/c42-36(41-39(27-14-4-1-5-15-27,28-16-6-2-7-17-28)29-18-8-3-9-19-29)25-24-35(37(43)44)40-38(45)46-26-34-32-22-12-10-20-30(32)31-21-11-13-23-33(31)34/h1-23,34-35H,24-26H2,(H,40,45)(H,41,42)(H,43,44)/t35-/m0/s1
InChI 密鑰
WDGICUODAOGOMO-DHUJRADRSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Fmoc-Gln(Trt)-OH 也称 Nα-Fmoc-Nδ-三苯甲基-L-谷氨酰胺,它是 Fmoc 保护的氨基酸衍生物,可用作合成肽的试剂。 由于谷氨酰胺受到三苯甲基保护,因此可以防止在反应中出现不需要的取代产物。
應用
Fmoc-Gln(Trt)-OH可用作Fmoc 固相肽合成的原料,用于合成fellutamide B 和 fellutamide B的N-辛酰基类似物。
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 1
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
Protection of carboxamide functions by the trityl residue. Application to peptide synthesis
P Sieber, et.al.
Tetrahedron Letters, 32, 739-742 (1991)
Neurotrophic peptide aldehydes: Solid phase synthesis of fellutamide B and a simplified analog
JS Schneekloth
Bioorganic & Medicinal Chemistry Letters, 16, 3855-3858 (2006)
Cathleen Jendrny et al.
Chembiochem : a European journal of chemical biology, 17(8), 719-726 (2015-11-18)
Serpin proteins irreversibly inhibit serine proteases, but only a small part of the serpin reactive-center loop (RCL) is responsible for the initial protein-protein interaction (PPI). To develop peptidic protease inhibitors, kallikrein-related peptidases 7 (KLK7) and 5 (KLK5) were chosen. Firstly
Marcus Pickhardt et al.
Current Alzheimer research, 14(7), 742-752 (2017-02-06)
Anti-aggregation drugs play an important role in therapeutic approaches for Alzheimer's disease. We have previously developed a number of compounds that are able to inhibit the pathological aggregation of Tau protein. One common obstacle to application is the limited penetration
Fatemeh Zabihi et al.
Journal of controlled release : official journal of the Controlled Release Society, 242, 35-41 (2016-07-30)
Nanogels offer many unique features rendering them as very attractive candidates for drug delivery. However, for their applications the loading capacity and specific encapsulation, in particular for hydrophobic drugs, in a complex media are two critical factors. In this work
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门