663107
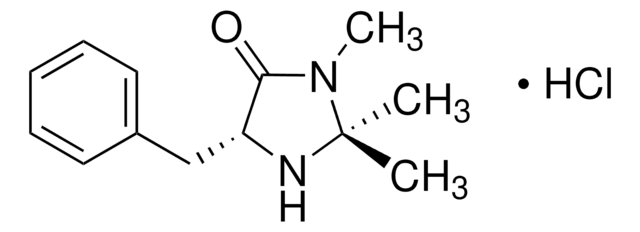
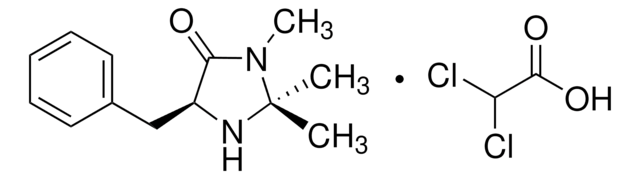
(2S,5S)-(−)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone
97%
Synonyme(s) :
(2S,5S)-2-tert-Butyl-3-methyl-5-phenylmethyl-4-imidazolidinone, (2S,5S)-5-Benzyl-2-tert-butyl-3-methyl-4-imidazolidinone
About This Item
Produits recommandés
Pureté
97%
Forme
solid
Pf
93-100 °C (lit.)
Groupe fonctionnel
phenyl
Chaîne SMILES
CN1[C@H](N[C@@H](Cc2ccccc2)C1=O)C(C)(C)C
InChI
1S/C15H22N2O/c1-15(2,3)14-16-12(13(18)17(14)4)10-11-8-6-5-7-9-11/h5-9,12,14,16H,10H2,1-4H3/t12-,14-/m0/s1
Clé InChI
SKHPYKHVYFTIOI-JSGCOSHPSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
- The chiral transformation reaction, including Friedel-Crafts and Mukaiyama-Michael reactions.
- The preparation of substituted spiroundecenetriones via asymmetric domino Knoevenagel/Diels-Alder reactions.
- The asymmetric synthesis of β-hydroxy aldehydes and their dimethylacetals via aldehyde-aldehyde aldol condensation reaction.
- The enantioselective α-fluorination of aldehydes using N-fluorobenzenesulfonamide as a fluorinating agent.
- The stereoselective preparation of (oxomethyl)oxabicyclo[3.2.1]octenones and tricyclic pyrroles via [4+3] cycloaddition of (trialkylsiloxy)pentadienals to furans.
Caractéristiques et avantages
- Superior enantiocontrol in numerous transformations
- High activities at low catalyst loadings
- Extraordinary functional group tolerance
Informations légales
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Articles
Discover Professor David MacMillan's groundbreaking metal-free asymmetric catalysis using imidazolidinone-based organocatalysts for versatile transformations.
Discover Professor David MacMillan's groundbreaking metal-free asymmetric catalysis using imidazolidinone-based organocatalysts for versatile transformations.
Discover Professor David MacMillan's groundbreaking metal-free asymmetric catalysis using imidazolidinone-based organocatalysts for versatile transformations.
Discover Professor David MacMillan's groundbreaking metal-free asymmetric catalysis using imidazolidinone-based organocatalysts for versatile transformations.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique
![(S)-α,α-Bis[3,5-bis(trifluoromethyl)phenyl]-2-pyrrolidinemethanol trimethylsilyl ether 97%](/deepweb/assets/sigmaaldrich/product/structures/396/398/09a397b1-b5f5-420f-98da-adf9017cef56/640/09a397b1-b5f5-420f-98da-adf9017cef56.png)


