P-050
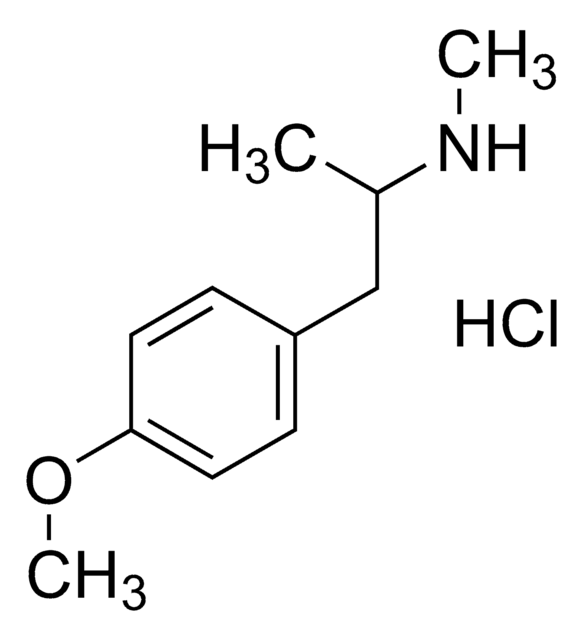
PMA hydrochloride solution
1.0 mg/mL in methanol (as free base), ampule of 1 mL, certified reference material, Cerilliant®
Sinónimos:
p-Methoxyamphetamine hydrochloride
About This Item
Productos recomendados
grado
certified reference material
formulario
liquid
Características
Snap-N-Spike®/Snap-N-Shoot®
envase
ampule of 1 mL
fabricante / nombre comercial
Cerilliant®
drug control
Narcotic Licence Schedule D (Switzerland); psicótropo (Spain); Decreto Lei 15/93: Tabela IIA (Portugal)
concentración
1.0 mg/mL in methanol (as free base)
técnicas
gas chromatography (GC): suitable
liquid chromatography (LC): suitable
aplicaciones
forensics and toxicology
formato
single component solution
temp. de almacenamiento
2-8°C
InChI
1S/C10H15NO.ClH/c1-8(11)7-9-3-5-10(12-2)6-4-9;/h3-6,8H,7,11H2,1-2H3;1H
Clave InChI
VSHZXOLHFRVZND-UHFFFAOYSA-N
Descripción general
Información legal
Producto relacionado
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Flam. Liq. 2 - STOT SE 1
Órganos de actuación
Eyes
Código de clase de almacenamiento
3 - Flammable liquids
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
49.5 °F - closed cup
Punto de inflamabilidad (°C)
9.7 °C - closed cup
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico