SAE0093
Beta-1,4-galactosyltransferase 1
B4GALT1 human recombinant, expressed in HEK 293 cells, 2000 units/mg protein
Synonyma:
Beta-1,4-GalTase 1, Beta4Gal-T1, UDP-Gal:beta-GlcNAc beta-1,4-galactosyltransferase 1, UDP-galactose:beta-N-acetylglucosamine beta-1,4-galactosyltransferase 1, b4Gal-T1
About This Item
Doporučené produkty
recombinant
expressed in HEK 293 cells
assay
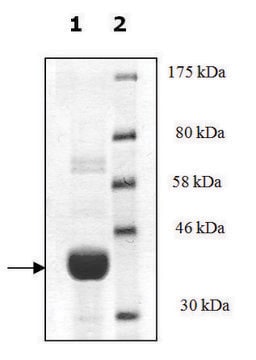
95% (SDS-PAGE)
form
lyophilized powder
specific activity
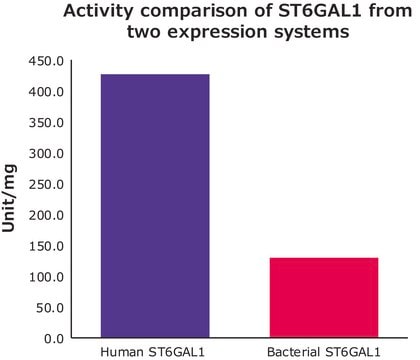
2000 units/mg protein
shipped in
ambient
storage temp.
−20°C
General description
Application
Biochem/physiol Actions
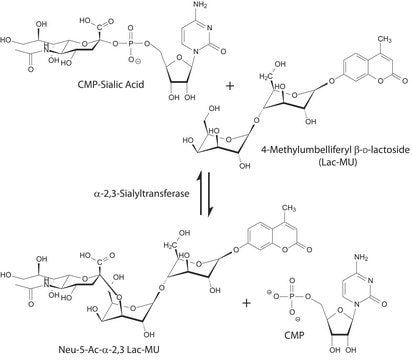
A major function of B4GALT1 is the addition of β(1→4) linked galactose residues to oligosaccharide acceptors with terminal N-acetylglucosamine residues. This is a late elongation step in the N-glycan processing pathway.B4GALT1 enzymatic activity is widely distributed in the vertebrate kingdom, in both mammals and non-mammals, including avians and amphibians.B4GALT1 enzymatic activity has also been demonstrated in a subset of plants which diverged from animals an estimated 1 billion years ago.B4GALT1 interacts with α-lactalbumin (LA), a protein expressed in the mammary gland during lactation, to form the lactose synthase (LS) complex that transfers galactose from UDP-α-D-Gal to glucose, producing the lactose secreted in milk.Defects in B4GALT1 are the cause of congenital disorder of glycosylation type 2D (CDG2D).Glomerular B4GALT1 expression has been found to be increased in IgA nephropathy. IgA binding and IgA-induced mesangial cell phosphorylation of spleen tyrosine kinase and IL-6 synthesis were inhibited by a panel of β(1→4) galactosyltransferase-specific antibodies, which suggests that IgA binds to the catalytic domain of β(1→4) galactosyltransferase.
Unit Definition
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Osvědčení o analýze (COA)
Vyhledejte osvědčení Osvědčení o analýze (COA) zadáním čísla šarže/dávky těchto produktů. Čísla šarže a dávky lze nalézt na štítku produktu za slovy „Lot“ nebo „Batch“.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Zákazníci si také prohlíželi
Sortimentní položky
Explore tools for glycosyltransferase synthesis and modification of glycans, such as glycosyltransferases and nucleotide sugar donors.
Enzymatic glycosyltransferase specificity challenges the one enzyme-one linkage concept.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.