E6412
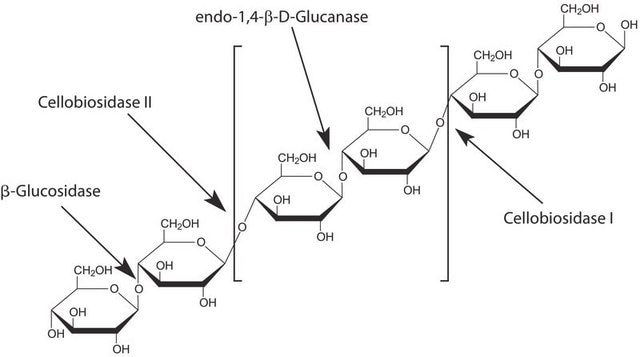
Cellobiohydrolase I from Hypocrea jecorina
0.13 U/mg, recombinant, expressed in corn
Synonyma:
Cel7A, Cellobiosidase, Cellulase
About This Item
Doporučené produkty
recombinant
expressed in corn
form
liquid
specific activity
0.13 U/mg
greener alternative product characteristics
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
greener alternative category
shipped in
dry ice
storage temp.
−20°C
Související kategorie
General description
Application
Biochem/physiol Actions
Unit Definition
Physical form
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Osvědčení o analýze (COA)
Vyhledejte osvědčení Osvědčení o analýze (COA) zadáním čísla šarže/dávky těchto produktů. Čísla šarže a dávky lze nalézt na štítku produktu za slovy „Lot“ nebo „Batch“.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Zákazníci si také prohlíželi
Sortimentní položky
Uncover more about glycosaminoglycans and proteoglycans including the structure of glycosaminoglycans (GAGs), the different types of GAGs, and their functions.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.