PHR1390
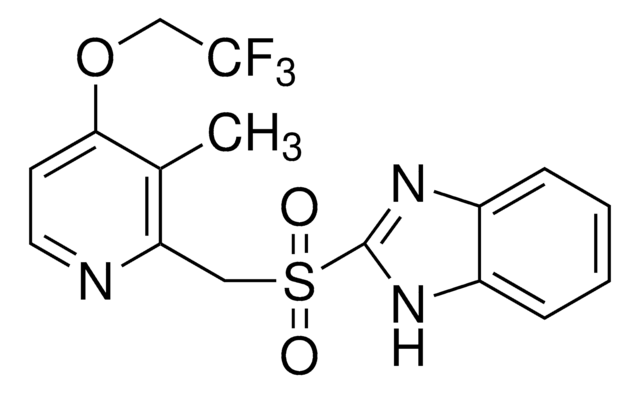
Lansoprazole
Pharmaceutical Secondary Standard; Certified Reference Material
Synonyma:
Lansoprazole, 2-[[[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl] methyl] sulfinyl]-1H-benzimidazole, LAN
About This Item
Doporučené produkty
grade
certified reference material
pharmaceutical secondary standard
Quality Level
agency
traceable to BP 872
traceable to Ph. Eur. Y0000773
traceable to USP 1356916
API family
lansoprazole
CofA
current certificate can be downloaded
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
SMILES string
Cc1c(CS(=O)c2nc3ccccc3[nH]2)nccc1OCC(F)(F)F
InChI
1S/C16H14F3N3O2S/c1-10-13(20-7-6-14(10)24-9-16(17,18)19)8-25(23)15-21-11-4-2-3-5-12(11)22-15/h2-7H,8-9H2,1H3,(H,21,22)
InChI key
MJIHNNLFOKEZEW-UHFFFAOYSA-N
Gene Information
human ... ATP4A(495) , ATP4B(496)
Hledáte podobné produkty? Navštivte Průvodce porovnáváním produktů
General description
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Application
Analysis Note
Other Notes
Footnote
Recommended products
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Vyberte jednu z posledních verzí:
Osvědčení o analýze (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumenty section.
Potřebujete-li pomoc, obraťte se na Zákaznická podpora
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Zákazníci si také prohlíželi
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.