79421
Phosphazene base P4-t-Bu solution
~0.8 M in hexane
Sinónimos:
1-tert-Butyl-4,4,4-tris(dimethylamino)-2,2-bis[tris(dimethylamino)-phosphoranylidenamino]-2λ5,4λ5-catenadi(phosphazene)
About This Item
Productos recomendados
form
liquid
Quality Level
concentration
~0.8 M in hexane
density
0.850-0.875 g/mL at 20 °C
SMILES string
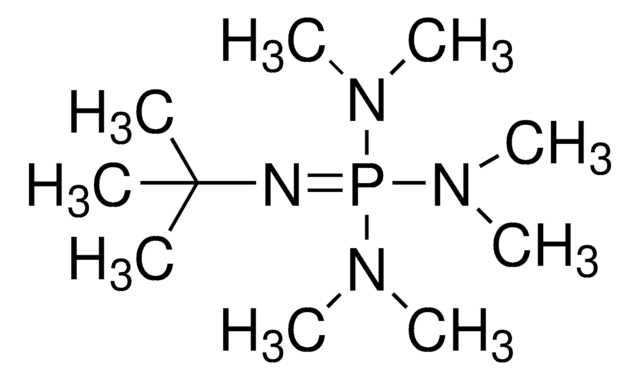
CN(C)P(=NP(=NC(C)(C)C)(N=P(N(C)C)(N(C)C)N(C)C)N=P(N(C)C)(N(C)C)N(C)C)(N(C)C)N(C)C
InChI
1S/C22H63N13P4/c1-22(2,3)23-36(24-37(27(4)5,28(6)7)29(8)9,25-38(30(10)11,31(12)13)32(14)15)26-39(33(16)17,34(18)19)35(20)21/h1-21H3
InChI key
NSRBCQCXZAYQHF-UHFFFAOYSA-N
Caution
Other Notes
signalword
Danger
Hazard Classifications
Aquatic Chronic 3 - Asp. Tox. 1 - Eye Dam. 1 - Flam. Liq. 2 - Repr. 2 - Skin Corr. 1B - STOT RE 1 Inhalation - STOT SE 3
target_organs
Central nervous system, Nervous system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
-14.8 °F - closed cup
flash_point_c
-26 °C - closed cup
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Artículos
Phosphazene base reagents are available as monomeric (P1 and BEMP), dimeric (P2), and tetrameric (P4) bases with different side chains to control their sterical hindrance.
Phosphazene base reagents are available as monomeric (P1 and BEMP), dimeric (P2), and tetrameric (P4) bases with different side chains to control their sterical hindrance.
Phosphazene base reagents are available as monomeric (P1 and BEMP), dimeric (P2), and tetrameric (P4) bases with different side chains to control their sterical hindrance.
Phosphazene base reagents are available as monomeric (P1 and BEMP), dimeric (P2), and tetrameric (P4) bases with different side chains to control their sterical hindrance.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico




![1,8-Diazabiciclo[5.4.0]undec-7-eno 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)






