647292
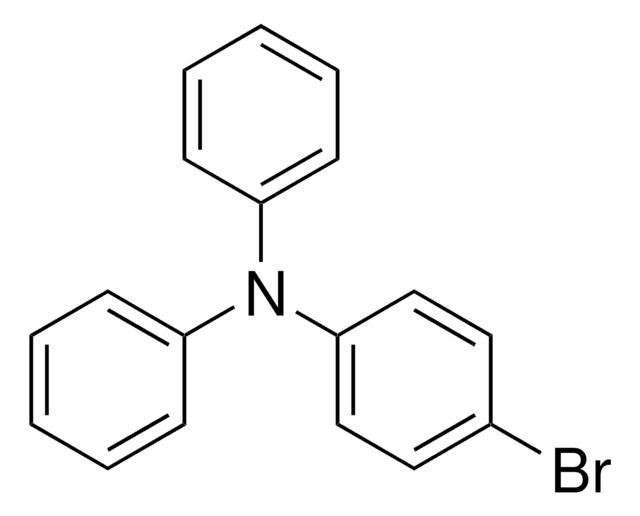
4-(Diphenylamino)phenylboronic acid
≥95%
Sinónimos:
4-(N,N-Diphenylamino)-1-phenylboronic acid, 4-(N,N-Diphenylamino)phenylboronic acid, 4-(N-Diphenylamino)phenylboronic acid, 4-(Diphenylamino)benzeneboronic acid, Triphenylamine-4-boronic acid
About This Item
Productos recomendados
assay
≥95%
form
powder
mp
110-115 °C (lit.)
SMILES string
OB(O)c1ccc(cc1)N(c2ccccc2)c3ccccc3
InChI
1S/C18H16BNO2/c21-19(22)15-11-13-18(14-12-15)20(16-7-3-1-4-8-16)17-9-5-2-6-10-17/h1-14,21-22H
InChI key
TWWQCBRELPOMER-UHFFFAOYSA-N
Categorías relacionadas
Application
- Strong multiphoton-excited blue photoluminescence and lasing from ladder-type oligo(p-phenylene)s
- Suzuki coupling reactions
- Ligand-free Suzuki reaction
Reagent used in Preparation of
- Push-pull arylvinyldiazine chromophores
- Benzothiadiazole-based fluorophores contg. triphenylamine functionality
- Blue light-emitting and hole-transporting materials for electroluminescent devices
- p-quaterphenyls laterally substituted with dimesitylboryl group for use as solid-state blue emitters
- Efficient sensitizers for dye-sensitized solar cells
- Prange electroluminescent materials for single-layer white polymer OLEDs
- Eeep-blue organic light emitting devices (OLEDs)
- Ligands for Organic Photovoltaic cells
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Self-healing soft electronic materials offer potential cost savings and reduced electronic waste.
Self-healing soft electronic materials offer potential cost savings and reduced electronic waste.
Self-healing soft electronic materials offer potential cost savings and reduced electronic waste.
Self-healing soft electronic materials offer potential cost savings and reduced electronic waste.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico![B-[4-(1,2,2-Triphenylethenyl)phenyl]boronic acid](/deepweb/assets/sigmaaldrich/product/structures/121/044/864e0829-e1de-4170-aae4-16c2b3ce4111/640/864e0829-e1de-4170-aae4-16c2b3ce4111.png)





![Benzo[b]thien-2-ylboronic acid ≥95%](/deepweb/assets/sigmaaldrich/product/structures/251/077/d0ead874-b533-4dcb-890d-8816a0018ccd/640/d0ead874-b533-4dcb-890d-8816a0018ccd.png)