115819
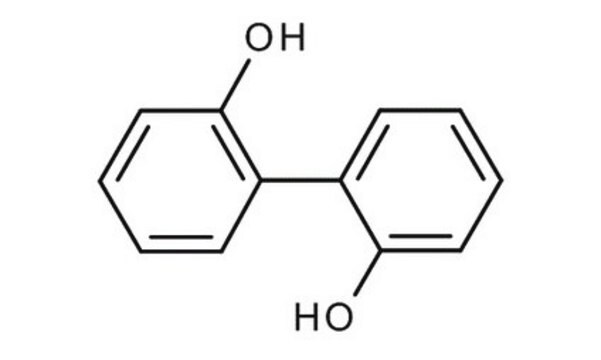
2,2′-Biphenol
99%
Sinónimos:
2,2′-Biphenyldiol, 2,2′-Dihydroxybiphenyl, 2,2′-Diphenol
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
HOC6H4C6H4OH
Número de CAS:
Peso molecular:
186.21
Beilstein:
1638363
Número CE:
Número MDL:
Código UNSPSC:
12162002
ID de la sustancia en PubChem:
NACRES:
NA.23
Productos recomendados
Ensayo
99%
Formulario
solid
bp
315 °C (lit.)
mp
108-110 °C (lit.)
cadena SMILES
Oc1ccccc1-c2ccccc2O
InChI
1S/C12H10O2/c13-11-7-3-1-5-9(11)10-6-2-4-8-12(10)14/h1-8,13-14H
Clave InChI
IMHDGJOMLMDPJN-UHFFFAOYSA-N
Categorías relacionadas
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 2
Punto de inflamabilidad (°F)
309.2 °F - closed cup - (External MSDS)
Punto de inflamabilidad (°C)
154 °C - closed cup - (External MSDS)
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
V V Subrahmanyam et al.
Xenobiotica; the fate of foreign compounds in biological systems, 20(12), 1369-1378 (1990-12-01)
1. 14C-Phenol was metabolized by rat bone marrow homogenate and H2O2. The homogenate catalyst, however, was inactivated by preincubation with H2O2, presumably due to inactivation of the enzyme(s) involved in phenol metabolism. 2. The majority of the metabolized 14C-phenol was
Zwe-Ling Kong et al.
Bioorganic & medicinal chemistry letters, 15(1), 163-166 (2004-12-08)
The neolignans, magnolol 1 and honokiol 2 have been reported to inhibit the growth of several tumor cell lines in vitro and in vivo. The chemical structure of magnolol and honokiol consists of biphenyl skeleton with phenolic and allylic functionalities.
Atsushi Kuwahara et al.
The Journal of organic chemistry, 70(2), 413-419 (2005-01-18)
The double N-arylation of primary amines with 2,2'-biphenylylene ditriflates was investigated for the synthesis of multisubstituted carbazoles. Palladium complexes supported by 2-dicyclohexylphosphino-2'-methylbiphenyl or Xantphos [4,5-bis(diphenylphosphino)-9,9-dimethylxanthene] were found to be efficient catalysts for the reaction. The catalysts allow the use of
Zengqi Xie et al.
Organic letters, 12(14), 3204-3207 (2010-06-22)
Facile nucleophilic substitution of two chlorine atoms by 2,2'-biphenol at one of the two bay areas (1,12- and 6,7-positions) of core-tetrachlorinated perylene bisimide afforded a novel, completely desymmetrized perylene bisimide building block, which could be further functionalized by substitution of
Bernd Schmidt et al.
The Journal of organic chemistry, 78(17), 8680-8688 (2013-08-01)
User-friendly protocols for the protecting group-free synthesis of 2,2'-biphenols via Suzuki-Miyaura coupling of o-halophenols and o-boronophenol are presented. The reactions proceed in water in the presence of simple additives such as K2CO3, KOH, KF, or TBAF and with commercially available
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico