E3763
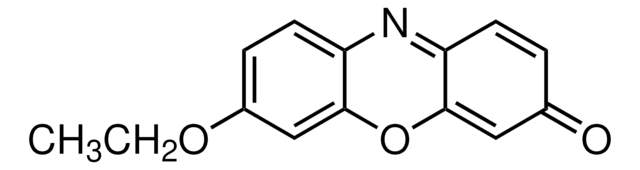
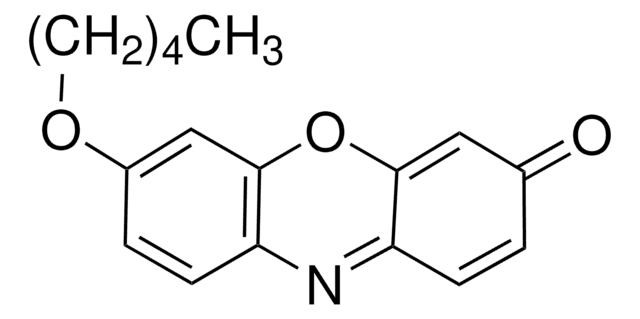
Resorufin ethyl ether
≥98% (TLC), powder
Sinónimos:
7-Ethoxy-3H-phenoxazin-3-one, Ethoxyresorufin, O7-Ethylresorufin
About This Item
Productos recomendados
Nivel de calidad
Ensayo
≥98% (TLC)
Formulario
powder
condiciones de almacenamiento
(Tightly closed. Dry)
técnicas
activity assay: suitable
color
orange to red
mp
223-225 °C (lit.)
solubilidad
chloroform: 9.80-10.20 mg/mL, clear, orange
idoneidad
suitable for fluorescence
temp. de almacenamiento
−20°C
cadena SMILES
CCOc1ccc2N=C3C=CC(=O)C=C3Oc2c1.Fc4c(F)c(F)c(OC(=O)CNC(=O)OCC5c6ccccc6-c7ccccc57)c(F)c4F
InChI
1S/C23H14F5NO4.C14H11NO3/c24-17-18(25)20(27)22(21(28)19(17)26)33-16(30)9-29-23(31)32-10-15-13-7-3-1-5-11(13)12-6-2-4-8-14(12)15;1-2-17-10-4-6-12-14(8-10)18-13-7-9(16)3-5-11(13)15-12/h1-8,15H,9-10H2,(H,29,31);3-8H,2H2,1H3
Clave InChI
ZOSYTBPPLWBBKM-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Research area: Cell Signaling
Aplicación
Sustratos
sustrato
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Phase I biotransformation reactions increase drug compound polarity, mainly occurring in hepatic circulation.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico