391573
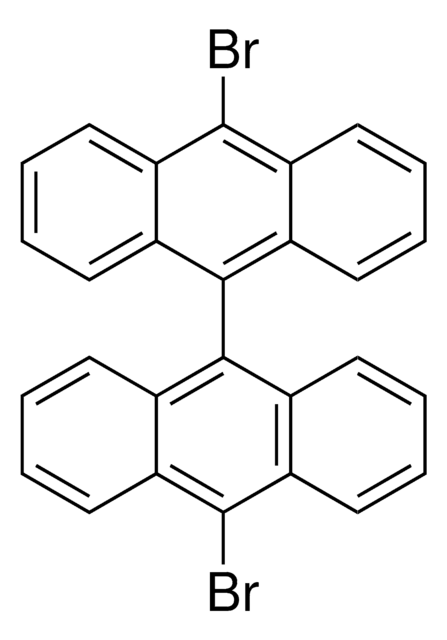
1-Bromopyrene
96%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C16H9Br
Número de CAS:
Peso molecular:
281.15
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
96%
Formulario
powder
mp
102-105 °C (lit.)
grupo funcional
bromo
cadena SMILES
Brc1ccc2ccc3cccc4ccc1c2c34
InChI
1S/C16H9Br/c17-14-9-7-12-5-4-10-2-1-3-11-6-8-13(14)16(12)15(10)11/h1-9H
Clave InChI
HYGLETVERPVXOS-UHFFFAOYSA-N
Descripción general
1-Bromopyrene, a polycyclic aromatic hydrocarbon (PAH), is a mono bromo substituted pyrene derivative. Its synthesis has been reported. Its gas phase UV-absorption spectra in the UV wavelength range at elevated temperature has been studied. Electron affinitiy (EA) of 1-bromopyrene has been investigated using electron attachment desorption chemical ionization mass spectrometry (DCI-MS) and triple quadrupole tandem mass spectrometry. It participates in the synthesis of novel ruthenium (II) bipyridine or terpyridine complexes bearing pyrene moiety. The reaction of 1-bromopyrene cation radical with water in acetonitrile has been analyzed using the electron transfer stopped-flow (ETSF) method.
Aplicación
1-Bromopyrene is suitable reagent used in the comparative study of effect of substituents of some pyrene derivatives in inducing phototoxicity, DNA damage and repair in human skin keratinocytes and light-induced lipid peroxidation in methanol. It is suitable reagent used in the study to investigate the UV photon-assisted thermal decomposition of PAHs at elevated temperature.
1-Bromopyrene may be used as a standard to compare its spectral properties with that of pyrene based fluorescence probe. It may be used to study the effects of the addition of halogen hetero-atoms on the vapor pressures and thermodynamics of polycyclic aromatic hydrocarbons.
It may be used in the synthesis of the following:
1-Bromopyrene may be used as a standard to compare its spectral properties with that of pyrene based fluorescence probe. It may be used to study the effects of the addition of halogen hetero-atoms on the vapor pressures and thermodynamics of polycyclic aromatic hydrocarbons.
It may be used in the synthesis of the following:
- 2-methyl-4-pyren-1-yl-but-3-yn-2-ol
- 1-ethynylpyrene
- silsesquioxane (SSQ) based hybrid
- ruthenium nanoparticles functionalized with pyrene moiety
- mono- and di-pyrenyl perfluoroalkanes
- oligo(1-bromopyrene)(OBrP) films
- dinitropyrene-derived DNA adduct
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Jillian L Goldfarb et al.
The Journal of chemical thermodynamics, 40(3), 460-466 (2008-03-01)
Knowledge of vapor pressures of high molar mass organics is essential to predicting their behavior in combustion systems as well as their fate and transport within the environment. This study involved polycyclic aromatic compounds (PACs) containing halogen hetero-atoms, including bromine
Gas-phase UV spectroscopy of anthracene, xanthone, pyrene, 1-bromopyrene and 1, 2, 4-trichlorobenzene at elevated temperatures.
Thony A and Rossi MJ.
Journal of Photochemistry and Photobiology A: Chemistry, 104(1), 25-33 (1997)
UV photon-assisted incineration of polycyclic aromatic hydrocarbons at elevated temperatures between 150 and 800?C.
Thony A and Rossi MJ.
Journal of Photochemistry and Photobiology A: Chemistry, 109(3), 267-280 (1997)
Syntheses of mono-and di-pyrenyl perfluoroalkanes.
Wiedenfeld D, et al.
Journal of Fluorine Chemistry, 104(2), 303-306 (2000)
Shu-Wen Yang et al.
The journal of physical chemistry. B, 109(35), 16628-16635 (2006-07-21)
An attempt to tune the electronic properties of pyrene (Py) by coupling it with a strong electron donor (-PhNMe2, DMA)/acceptor (anthronitrile, AN) through an ethynyl bridge has been undertaken. A moderate electron donor (iPrOPh-, IPP)/acceptor (2-quinolinyl, 2Q) has also been
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico