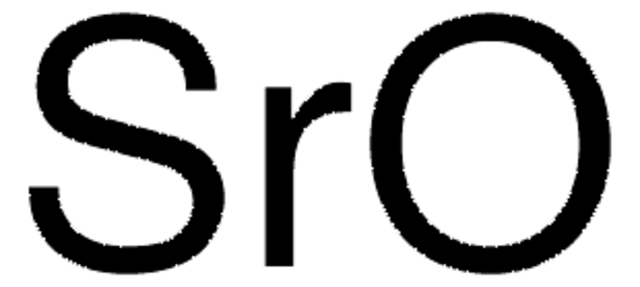227692
Sulfur trioxide
contains stabilizer, ≥99%
Sinonimo/i:
Sulfuric anhydride
About This Item
Prodotti consigliati
Densità del vapore
2.8 (vs air)
Tensione di vapore
280 mmHg ( 25 °C)
Saggio
≥99%
Forma fisica
liquid
contiene
stabilizer
Impiego in reazioni chimiche
reagent type: oxidant
Impurezze
low-melting solid polymer (the formation of which is unavoidable)
P. eboll.
44.7 °C (lit.)
Punto di fusione
16.8 °C (lit.)
Densità
1.97 g/mL at 25 °C (lit.)
Stringa SMILE
O=S(=O)=O
InChI
1S/O3S/c1-4(2)3
AKEJUJNQAAGONA-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Skin Corr. 1A - STOT SE 3
Organi bersaglio
Respiratory system
Rischi supp
Codice della classe di stoccaggio
8B - Non-combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Faceshields, Gloves, Goggles
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Which document(s) contains shelf-life or expiration date information for a given product?
If available for a given product, the recommended re-test date or the expiration date can be found on the Certificate of Analysis.
How do I get lot-specific information or a Certificate of Analysis?
The lot specific COA document can be found by entering the lot number above under the "Documents" section.
How do I find price and availability?
There are several ways to find pricing and availability for our products. Once you log onto our website, you will find the price and availability displayed on the product detail page. You can contact any of our Customer Sales and Service offices to receive a quote. USA customers: 1-800-325-3010 or view local office numbers.
Is Product 227692, Sulfur trioxide, OK to use if it contains solids?
This product may contain low-melting solid polymer, which is unavoidable. If the material is not completely liquid, place the bottle in a glass beaker or metal pail and warm under heat lamp or in an oven at 35°C. DO NOT OVERHEAT! This product boils at 44.7°C. Melting may take up to one week. When the material is liquid, it is ready for use.
What is the stabilizer used in Product 227692, Sulfur trioxide?
The manufacturer considers the identity of the stabilizer to be proprietary information.
Why does Product 227692, Sulfur trioxide, contain a stabilizer?
The stabilizer is present to facilitate melting, not to prevent polymerization.
How is Product 227692, Sulfur trioxide, packaged?
This product is packaged in a screw-capped glass bottle.
What is the Department of Transportation shipping information for this product?
Transportation information can be found in Section 14 of the product's (M)SDS.To access the shipping information for this material, use the link on the product detail page for the product.
My question is not addressed here, how can I contact Technical Service for assistance?
Ask a Scientist here.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.