261432
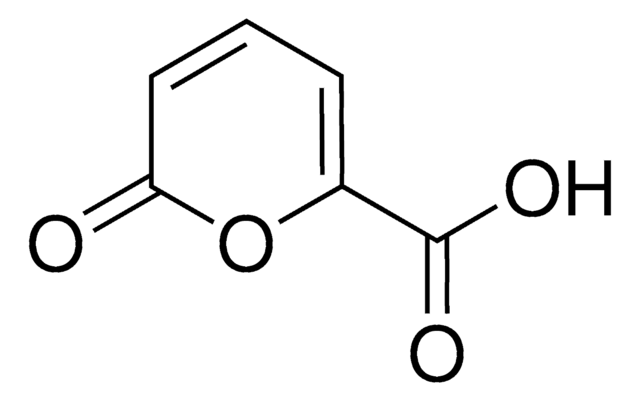
Methyl coumalate
98%
Sinônimo(s):
Methyl 2-oxo-2H-pyran-5-carboxylate, Methyl 2-pyrone-5-carboxylate
About This Item
Produtos recomendados
Ensaio
98%
forma
solid
pb
178-180 °C/60 mmHg (lit.)
pf
65-67 °C (lit.)
cadeia de caracteres SMILES
COC(=O)C1=COC(=O)C=C1
InChI
1S/C7H6O4/c1-10-7(9)5-2-3-6(8)11-4-5/h2-4H,1H3
chave InChI
HHWWWZQYHPFCBY-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Artigos
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica