H4405
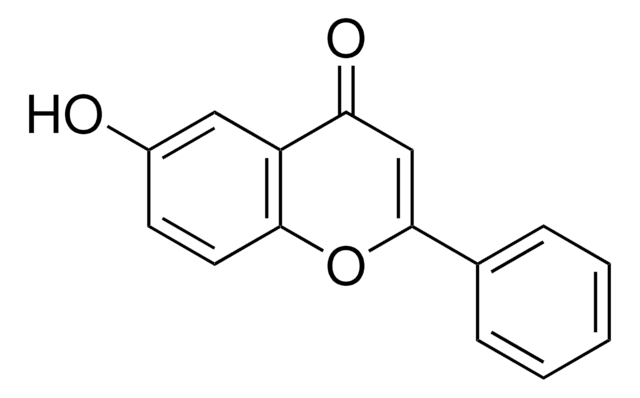
5-Hydroxyflavone
≥97%
Synonyme(s) :
5-Hydroxy-2-phenylchromone, NSC 26745, Primuletin
About This Item
Produits recommandés
Pureté
≥97%
Chaîne SMILES
Oc1cccc2OC(=CC(=O)c12)c3ccccc3
InChI
1S/C15H10O3/c16-11-7-4-8-13-15(11)12(17)9-14(18-13)10-5-2-1-3-6-10/h1-9,16H
Clé InChI
IYBLVRRCNVHZQJ-UHFFFAOYSA-N
Informations sur le gène
rat ... Gabra2(29706)
Application
- Condensation reactions for synthesis of copper(II) complexes as bioactive molecules to combat antioxidants
- Thermal behavior studies of vanadyl complexes with flavone derivatives in terms of insulin-mimetic agents
- O-methylation with di-Me carbonate
- DFT studies on excited-state intramolecular proton transfer
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique