Größe auswählen
€ 104,00
€ 351,05
Über diesen Artikel
Fortfahren mit
Produktname
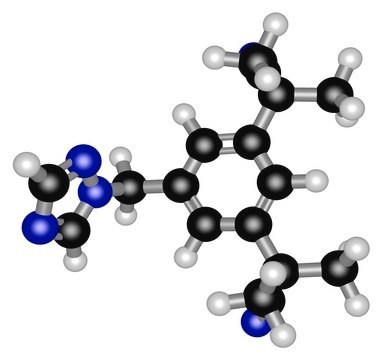
Anastrozol, ≥98% (HPLC)
SMILES string
[n]2(ncnc2)Cc1cc(cc(c1)C(C)(C)C#N)C(C)(C)C#N
InChI
1S/C17H19N5/c1-16(2,9-18)14-5-13(8-22-12-20-11-21-22)6-15(7-14)17(3,4)10-19/h5-7,11-12H,8H2,1-4H3
InChI key
YBBLVLTVTVSKRW-UHFFFAOYSA-N
assay
≥98% (HPLC)
form
solid
solubility
DMSO: 40 mg/mL
originator
AstraZeneca
storage temp.
room temp
Quality Level
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
1 of 4
Dieser Artikel | |||
|---|---|---|---|
| form solid | form powder | form - | form powder |
| assay ≥98% (HPLC) | assay ≥98% (HPLC) | assay - | assay ≥98% (HPLC) |
| Quality Level 100 | Quality Level 100 | Quality Level 300 | Quality Level 100 |
| storage temp. room temp | storage temp. room temp | storage temp. 2-30°C | storage temp. room temp |
| solubility DMSO: 40 mg/mL | solubility DMSO: >15 mg/mL | solubility - | solubility DMSO: 5 mg/mL |
| originator AstraZeneca | originator - | originator - | originator - |
Application
- as a positive control in DNA fragmentation (ladder) assay[1]
- to investigate its effects along with extra virgin olive oil and its major fatty acid component (omega-9 OA) in estrogen receptor positive mammary adenocarcinoma cells[2]
- to study its effects on viability, cell proliferation and apoptosis in Glioblastoma multiforme model in vivo[3]
Biochem/physiol Actions
Features and Benefits
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Repr. 1B
Lagerklasse
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Active Filters
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung