Über diesen Artikel
Fortfahren mit
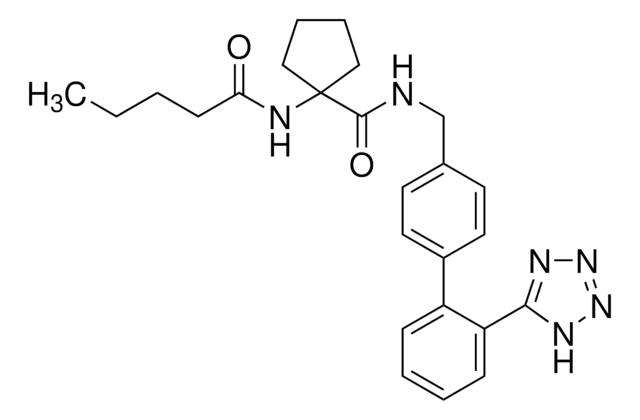
SMILES string
[nH]1nnnc1c2c(cccc2)c3ccc(cc3)C[n]4c5c(nc4CCC)C(OC5=O)(C)C
InChI
1S/C24H24N6O2/c1-4-7-19-25-21-20(23(31)32-24(21,2)3)30(19)14-15-10-12-16(13-11-15)17-8-5-6-9-18(17)22-26-28-29-27-22/h5-6,8-13H,4,7,14H2,1-3H3,(H,26,27,28,29)
InChI key
JUQNVWFXORBZQJ-UHFFFAOYSA-N
grade
certified reference material, pharmaceutical secondary standard
agency
traceable to USP 1478378
API family
olmesartan
CofA
current certificate can be downloaded
packaging
pkg of 40 mg
application(s)
pharmaceutical
format
neat
storage temp.
-10 to -25°C
Quality Level
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
1 of 4
Dieser Artikel | PHR1875 | PHR1958 | PHR2018 |
|---|---|---|---|
| application(s) pharmaceutical | application(s) pharmaceutical small molecule | application(s) pharmaceutical | application(s) pharmaceutical |
| format neat | format neat | format - | format neat |
| Quality Level 300 | Quality Level 300 | Quality Level 300 | Quality Level 300 |
| grade certified reference material, pharmaceutical secondary standard | grade certified reference material, pharmaceutical secondary standard | grade certified reference material, pharmaceutical secondary standard | grade certified reference material, pharmaceutical secondary standard |
| storage temp. -10 to -25°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| packaging pkg of 40 mg | packaging pkg of 30 mg | packaging pkg of 50 mg | packaging pkg of 20 mg |
General description
Application
Analysis Note
Other Notes
Ähnliches Produkt
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Active Filters
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung