D194255
1,3-Dinitrobenzol
97% anhydrous basis
Synonym(e):
1,3-Dinitrobenzene, DNB, Meta-dinitrobenzene, m-Dinitrobenzene
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
97% anhydrous basis
Form
solid
Verunreinigungen
≤10.0% H2O
bp
297 °C (lit.)
mp (Schmelzpunkt)
84-86 °C (lit.)
Dichte
1.368 g/mL at 25 °C (lit.)
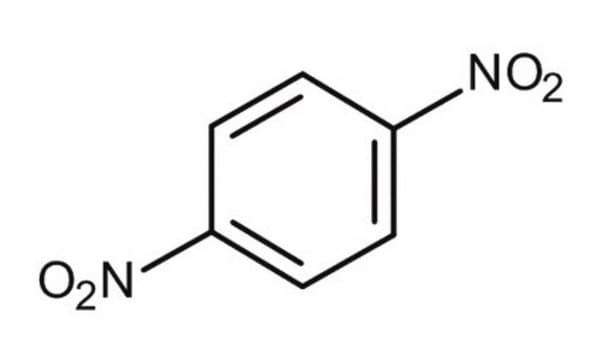
SMILES String
[O-][N+](=O)c1cccc(c1)[N+]([O-])=O
InChI
1S/C6H4N2O4/c9-7(10)5-2-1-3-6(4-5)8(11)12/h1-4H
InChIKey
WDCYWAQPCXBPJA-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Anwendung
- 2′,6′- dinitrobiphenyl-4-ol and 1-nitrodibenzofuran via copper-catalyzed regioselective cross-coupling reaction with 4-iodophenol and 2-iodophenol respectively.
- 1H-indazole derivatives by reacting with various N-tosylhydrazones in the presence of a base catalyst.
- 3-(1H-Tetrazol-1-yl)benzenamine by reacting with triethyl orthoformate and NaN3 in the presence of a three-functional redox catalytic system.
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Lagerklassenschlüssel
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flammpunkt (°F)
302.0 °F - closed cup
Flammpunkt (°C)
150 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.