Alle Fotos(1)
Wichtige Dokumente
178594
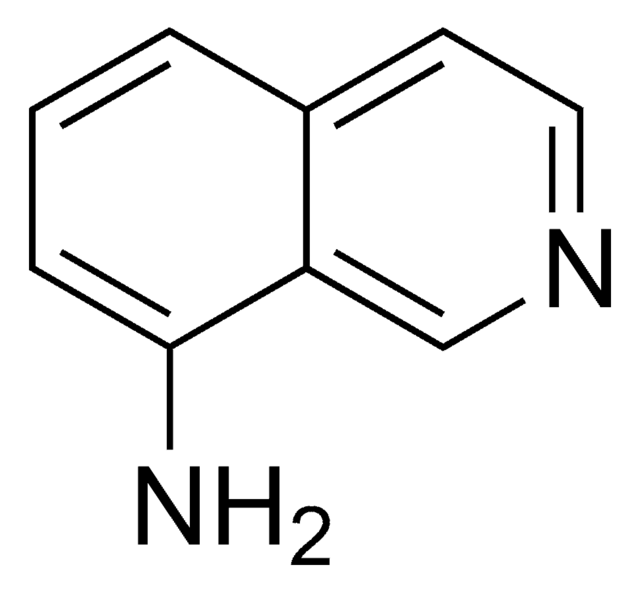
1-Aminoisochinolin
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C9H8N2
CAS-Nummer:
Molekulargewicht:
144.17
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
mp (Schmelzpunkt)
122-124 °C (lit.)
SMILES String
Nc1nccc2ccccc12
InChI
1S/C9H8N2/c10-9-8-4-2-1-3-7(8)5-6-11-9/h1-6H,(H2,10,11)
InChIKey
OSILBMSORKFRTB-UHFFFAOYSA-N
Anwendung
1-Aminoisoquinoline was used in the synthesis of pyrimidoisoquinolinone.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Persönliche Schutzausrüstung
dust mask type N95 (US), Eyeshields, Gloves
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Scott P Brown et al.
Journal of medicinal chemistry, 52(10), 3159-3165 (2009-04-24)
We apply a high-throughput formulation of the molecular mechanics with Poisson-Boltzmann surface area (htMM-PBSA) to estimate relative binding potencies on a set of 308 small-molecule ligands in complex with the proteins urokinase, PTP-1B, and Chk-1. We observe statistically significant correlation
Adrian L Smith et al.
Journal of medicinal chemistry, 52(20), 6189-6192 (2009-09-22)
The discovery and optimization of a novel series of aminoisoquinolines as potent, selective, and efficacious inhibitors of the mutant B-Raf pathway is presented. The N-linked pyridylpyrimidine benzamide 2 was identified as a potent, modestly selective inhibitor of the B-Raf enzyme.
[A practical method for the synthesis of 1-aminoisoquinoline].
S Giorgi-Renault et al.
Annales pharmaceutiques francaises, 41(6), 555-557 (1983-01-01)
Xiaohong Wei et al.
Organic letters, 13(17), 4636-4639 (2011-08-05)
[RhCp*Cl(2)](2) can catalyze the oxidative coupling of N-aryl and N-alkyl benzamidines with alkynes to give N-substituted 1-aminoisoquinolines in high selectivity.
Hervé Bibas et al.
The Journal of organic chemistry, 67(8), 2619-2631 (2002-04-13)
The synthesis, spectroscopic properties, and chemical reactions of the stable (neopentylimino)-, (mesitylimino)-, and (o-tert-butylphenylimino)propadienones (6) are reported. Nucleophilic addition of amines affords the malonic amidoamidines 7 and 8. 3,5-Dimethylpyrazole reacts analogously to form 9b. Addition of 1,2-dimethylhydrazine produces pyrazolinones 10-12.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.