Select a Size
| Pack Size | SKU | Availability | Price |
|---|
About This Item
Quality Level
assay
≥98.0% (HPLC)
form
powder or crystals
application(s)
food and beverages
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
2-8°C
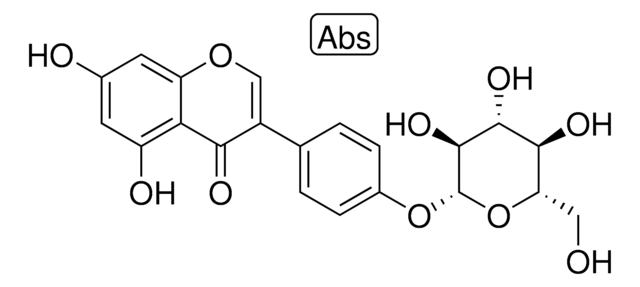
SMILES string
OC1O[C@H](COC(=O)c2cc(O)c(O)c(O)c2)[C@@H](O)[C@]1(O)COC(=O)c3cc(O)c(O)c(O)c3
InChI
1S/C20H20O14/c21-9-1-7(2-10(22)14(9)25)17(28)32-5-13-16(27)20(31,19(30)34-13)6-33-18(29)8-3-11(23)15(26)12(24)4-8/h1-4,13,16,19,21-27,30-31H,5-6H2/t13-,16-,19?,20-/m1/s1
InChI key
FEPAFOYQTIEEIS-IZUGRSKYSA-N
General description
Application
1 of 1
This Item | |||
|---|---|---|---|
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| application(s) food and beverages | application(s) food and beverages | application(s) food and beverages | application(s) metabolomics |
| assay ≥98.0% (HPLC) | assay ≥95.0% (HPLC) | assay ≥97.0% (HPLC) | assay ≥98% (HPLC) |
| form powder or crystals | form - | form - | form powder |
| Quality Level 100 | Quality Level - | Quality Level 100 | Quality Level 200 |
Still not finding the right product?
Explore all of our products under Hamamelitannin
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 188212-5G | 04061838758781 |
| 04646-5MG-F | 04061838635594 |