推薦產品
等級
pharmaceutical primary standard
API 家族
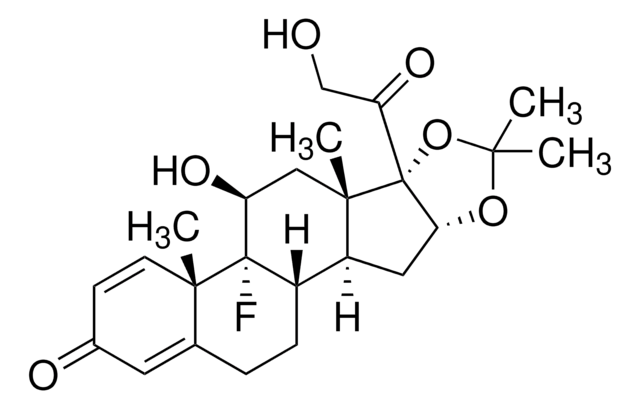
triamcinolone
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
SMILES 字串
[H][C@@]12CCC3=CC(=O)C=C[C@]3(C)[C@@]1(F)[C@@H](O)C[C@@]4(C)[C@@]2([H])C[C@H]5OC(C)(C)O[C@@]45C(=O)CO
InChI
1S/C24H31FO6/c1-20(2)30-19-10-16-15-6-5-13-9-14(27)7-8-21(13,3)23(15,25)17(28)11-22(16,4)24(19,31-20)18(29)12-26/h7-9,15-17,19,26,28H,5-6,10-12H2,1-4H3/t15-,16-,17-,19+,21-,22-,23-,24+/m0/s1
InChI 密鑰
YNDXUCZADRHECN-JNQJZLCISA-N
基因資訊
human ... NR3C1(2908)
尋找類似的產品? 前往 產品比較指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Triamcinolone acetonide for system suitability EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Repr. 1B
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Catherine McMillan et al.
The Journal of hand surgery, 39(10), 1942-1947 (2014-08-26)
To compare long-term outcomes and retreatment rates for patients with Dupuytren disease who underwent needle aponeurotomy (NA) combined with a series of triamcinolone acetonide injections or underwent NA alone as part of a prior randomized controlled trial. During this follow-up
Kent W Small et al.
Ophthalmology, 121(4), 952-958 (2014-02-11)
To report a series of cases with fungal endophthalmitis occurring after intravitreal injection of triamcinolone derived from a single lot prepared by a compounding pharmacy. Retrospective, observational case series. Seventeen eyes treated with triamcinolone obtained from a single lot subsequently
C M Jermak et al.
Survey of ophthalmology, 52(5), 503-522 (2007-08-28)
Triamcinolone acetonide has been effectively used in ocular therapeutics for over 50 years. Its use has increased dramatically in recent years for periocular and intraocular treatment of retinal vasculature disease and uveitis. This comprehensive review discusses the pharmacokinetics of triamcinolone
Taygan Yilmaz et al.
Ophthalmology, 116(5), 902-911 (2009-05-05)
To compare intravitreal triamcinolone acetonide (IVTA) injection versus no treatment or sub-Tenon triamcinolone acetonide (STTA) injection in improving visual acuity (VA) of patients with refractory diabetic macular edema (DME; unresponsive to focal laser therapy). Diabetic macular edema is the leading
Luis A Solchaga et al.
Journal of orthopaedic research : official publication of the Orthopaedic Research Society, 32(1), 145-150 (2013-09-11)
This study compared the effect of intra-tendon (IT) delivery of recombinant human platelet-derived growth factor-BB (rhPDGF-BB), platelet-rich plasma (PRP) and corticosteroids in a rat tendinopathy model. Seven days after collagenase induction of tendinopathy, a 30-µl IT injection was administered. Treatments
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務