推薦產品
等級
analytical standard
品質等級
化驗
≥90.0% (HPLC)
儲存期限
limited shelf life, expiry date on the label
技術
HPLC: suitable
gas chromatography (GC): suitable
應用
food and beverages
形式
neat
儲存溫度
2-8°C
SMILES 字串
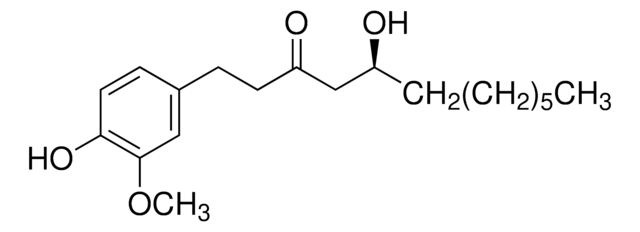
CCCCC\C=C\C(=O)CCc1ccc(O)c(OC)c1
InChI
1S/C17H24O3/c1-3-4-5-6-7-8-15(18)11-9-14-10-12-16(19)17(13-14)20-2/h7-8,10,12-13,19H,3-6,9,11H2,1-2H3/b8-7+
InChI 密鑰
OQWKEEOHDMUXEO-BQYQJAHWSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
應用
Refer to the product′s Certificate of Analysis for more information on a suitable instrument technique. Contact Technical Service for further support.
包裝
Bottomless glass bottle. Contents are inside inserted fused cone.
其他說明
This compound is commonly found in plants of the genus: zingiber
推薦產品
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
客戶也查看了
Yingdong Zhu et al.
PloS one, 8(1), e54677-e54677 (2013-02-06)
Our previous study found that [6]-shogaol, a major bioactive component in ginger, is extensively metabolized in cancer cells and in mice. It is unclear whether these metabolites retain bioactivity. The aim of the current study is to synthesize the major
Min-Ji Bak et al.
Molecules (Basel, Switzerland), 17(7), 8037-8055 (2012-07-06)
The rhizome of ginger (Zingiber officinale Roscoe) is known to have several bioactive compounds including gingerols and shogaols which possess beneficial health properties such as anti-inflammatory and chemopreventive effects. Based on recent observations that 6-shogaol may have more potent bioactivity
Qingliang Qiao et al.
Journal of chromatography. A, 1218(36), 6187-6190 (2011-01-05)
The flash high speed counter-current chromatographic (FHSCCC) separation of gingerols and 6-shogaol was performed on a HSCCC instrument equipped with a 1200-ml column (5 mm tubing i.d.) at a flow rate of 25 ml/min. The performance met the FHSCCC feature
Yanke Yu et al.
The AAPS journal, 13(3), 417-426 (2011-06-04)
Ginger extracts have been studied in various clinical trials for different indications. However, the pharmacokinetics of the ginger active constituents in human biological matrices is not well investigated. This study aims to develop a LC-MS/MS method for simultaneous measurement of
Sehwan Shim et al.
Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 50(3-4), 597-602 (2011-12-07)
[6]-Shogaol has beneficial effects in spinal neuronal regeneration, but associated molecules and mechanisms are not identified. Neurotrophic factors, including brain-derived neurotrophic factor (BDNF), are associated with proliferation and differentiation of neuronal cells and exert a neuroprotective effect in neurodegenerative models.
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務
![[6]-Gingerol, Zingiber officinale An antitumor, apoptosis-inducing compound of the ginger family that blocks EGF-induced cell transformation by inhibiting the activation of Activator Protein-1 (AP-1).](/deepweb/assets/sigmaaldrich/product/images/140/919/0846df46-0b67-4c28-b99d-87e177be65b2/640/0846df46-0b67-4c28-b99d-87e177be65b2.jpg)
![[10]-姜酮醇 ≥98% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/224/210/b4f3e699-03b9-4112-89c1-a63f196344d0/640/b4f3e699-03b9-4112-89c1-a63f196344d0.png)