推薦產品
品質等級
化驗
97%
形狀
powder or crystals
mp
127-132 °C
SMILES 字串
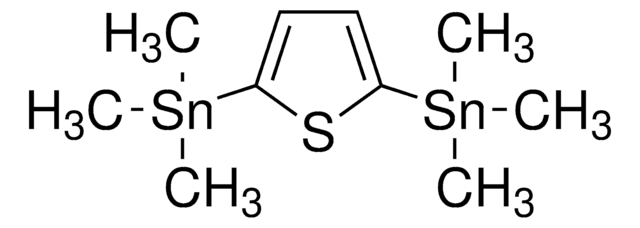
C[Sn](C)(C)c1cc2sc(cc2s1)[Sn](C)(C)C
InChI
1S/C6H2S2.6CH3.2Sn/c1-3-7-6-2-4-8-5(1)6;;;;;;;;/h1-2H;6*1H3;;
InChI 密鑰
HDZULVYGCRXVNQ-UHFFFAOYSA-N
一般說明
應用
2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene can be used as a copolymer in the synthesis of thiophene based materials for the fabrication of organic electronic devices such as organic field effect transistors (OFETs), organic thin film transistors(OTFTs) and organic photovoltaic cells (OPVs).[3][4]
2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene is a synthetic intermediate, which can be used in the synthesis of polythiophenes based fluorinated polymers for the optoelectronic applications.[5] It can also be used in the synthesis of PDBT-co-TT via Still coupling polymerization for the fabrication of organic thin film transistors (OTFTs)and photovoltaic devices.[2][3][1]
訊號詞
Danger
危險分類
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1
儲存類別代碼
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Dialkyl-Substituted Thieno [3, 2-b] thiophene-Based Polymers Containing 2, 2 `-Bithiophene, Thieno [3, 2-b] thiophene, and Ethynylene Spacers
San Miguel L and Matzger AJ
Macromolecules, 40(26), 9233-9237 (2007)
Thienoisoindigo-based copolymer with fused thieno (3, 2-b) thiophene as a donor in thin film transistor applications with high performance
Chen C, et al.
Journal of Material Chemistry C, 3(1), 33-36 (2015)
Using Molecular Design to Increase Hole Transport: Backbone Fluorination in the Benchmark Material Poly (2, 5-bis (3-alkylthiophen-2-yl) thieno [3, 2-b]-thiophene (pBTTT)
Boufflet P, et al.
Advances in Functional Materials, 25(45), 7038-7048 (2015)
A High Mobility P-Type DPP-Thieno [3, 2-b] thiophene Copolymer for Organic Thin-Film Transistors
Li Y, et al.
Advanced Materials, 22(43), 4862-4866 (2010)
Hugo Bronstein et al.
Journal of the American Chemical Society, 133(10), 3272-3275 (2011-02-22)
We report the synthesis and polymerization of a novel thieno[3,2-b]thiophene-diketopyrrolopyrrole-based monomer. Copolymerization with thiophene afforded a polymer with a maximum hole mobility of 1.95 cm(2) V(-1) s(-1), which is the highest mobility from a polymer-based OFET reported to date. Bulk-heterojunction
文章
Organic electronics promise renewable energy solutions surpassing silicon-based tech.
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務![1,1′-[4,8-Bis[5-(2-ethylhexyl)-2-thienyl]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl]bis[1,1,1-trimethylstannane]](/deepweb/assets/sigmaaldrich/product/structures/611/912/a638a6fe-ca7b-4674-8023-df4c0921a9fd/640/a638a6fe-ca7b-4674-8023-df4c0921a9fd.png)



![苯并[1,2B:4,5-B]二噻吩-4- -1,8-二酮 97%](/deepweb/assets/sigmaaldrich/product/structures/418/544/b7faac0b-ad09-4b42-a9fa-aeb38017a39e/640/b7faac0b-ad09-4b42-a9fa-aeb38017a39e.png)
![噻吩并[3,2-b]噻吩 95%](/deepweb/assets/sigmaaldrich/product/structures/353/609/429fd4bf-e217-4371-80a3-9e5a4d88908b/640/429fd4bf-e217-4371-80a3-9e5a4d88908b.png)



