全部照片(2)
About This Item
經驗公式(希爾表示法):
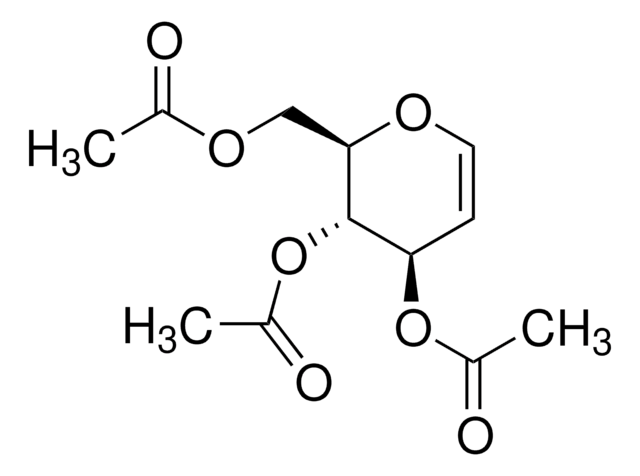
C6H10O4
CAS號碼:
分子量::
146.14
Beilstein:
81690
EC號碼:
MDL號碼:
分類程式碼代碼:
12352201
PubChem物質ID:
NACRES:
NA.22
推薦產品
化驗
95%
光學活性
[α]22/D −21.5°, c = 1.2 in methanol
mp
99-103 °C (lit.)
儲存溫度
2-8°C
SMILES 字串
OC[C@H]1OC=C[C@@H](O)[C@H]1O
InChI
1S/C6H10O4/c7-3-5-6(9)4(8)1-2-10-5/h1-2,4-9H,3H2/t4-,5-,6-/m1/s1
InChI 密鑰
YVECGMZCTULTIS-HSUXUTPPSA-N
應用
可同时用于寡糖的溶液相和固相合成的重要结构单元。
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Aditi Agarwal et al.
The Journal of organic chemistry, 69(18), 6137-6140 (2004-09-18)
Perchloric acid supported on silica gel acts as an excellent reagent system in converting glucals into 2,3-unsaturated-O-glucosides in good to excellent yields in short reaction time with good alpha selectivity. Primary, secondary, and allylic alcohols, phenols, and thiols react with
Chi-Li Chen et al.
Organic letters, 11(2), 409-412 (2009-01-09)
With focus on the steric effects present in the transition states for the [3,3]-sigmatropic rearrangement, the substrate 5 has been designed to improve the overall stereoselectivity of the Ireland-Claisen rearrangement. Experimentally, it has been found that (1) only Z-6 rearranges
Feiqing Ding et al.
Organic letters, 13(4), 652-655 (2011-01-20)
A highly efficient synthesis of L-ristosamine and L-epi-daunosamine glycosides via BF(3)·OEt(2) promoted tandem hydroamination/glycosylation of 3,4-di-O-acetyl-6-deoxy-L-glucal and L-galactal has been developed. The new method proceeds in a completely stereocontrolled manner within a short reaction time. Preparation of a library of
Jürgen Geiger et al.
The Journal of organic chemistry, 72(12), 4367-4377 (2007-05-17)
A 3,4-O-unprotected galactal derivative having bulky 6-O-TIPS protection (compound 2) could be regioselectively 3-O-glycosylated with O-(galactopyranosyl) trichloroacetimidates; depending on the protecting group pattern stereoselectively alpha- and beta-linked disaccharides were obtained. With O-(2-azido-2-deoxyglucopyransyl) trichloroacetimidate as donor (compound 10A), glycosylation of 2
L Kiss et al.
Carbohydrate research, 291, 43-52 (1996-09-23)
C-(2-Deoxy-D-lyxo-hex-1-enopyranosyl)formamide was prepared from acetylated C-(beta-D-galactopyranosyl)formamide by a radical-mediated bromination-zinc/N-methylimidazole-induced reductive elimination-Zemplén deacetylation reaction sequence. The preparation of acetylated 5-(2-deoxy-D-lyxo-hex-1-enopyranosyl)tetrazole was improved. Methyl C-(2-deoxy-D-lyxo-hex-1-enopyranosyl)formimidate was transformed by benzylamine into N-benzyl-C-(2-deoxy-D-lyxo-hex-1-enopyranosyl)formamidine and, after hydrolysis to methyl C-(2-deoxy-D-lyxo-hex-1-enopyranosyl)formate, into N-benzyl-C-(2-deoxy-D-lyxo-hex-1-enopyranosyl)formamide. A series
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務