全部照片(2)
About This Item
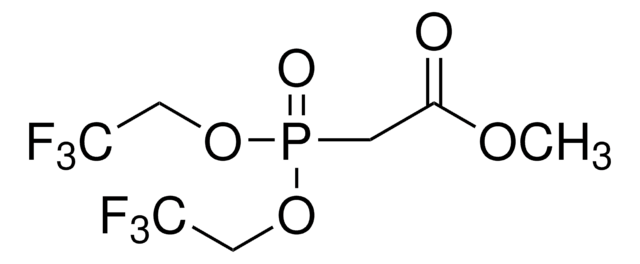
線性公式:
[(CF3)2CHO]3P
CAS號碼:
分子量::
532.06
Beilstein:
2315565
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推薦產品
化驗
98%
形狀
liquid
折射率
n20/D 1.300 (lit.)
bp
130 °C (lit.)
密度
1.69 g/mL at 25 °C (lit.)
SMILES 字串
FC(F)(F)C(OP(OC(C(F)(F)F)C(F)(F)F)OC(C(F)(F)F)C(F)(F)F)C(F)(F)F
InChI
1S/C9H3F18O3P/c10-4(11,12)1(5(13,14)15)28-31(29-2(6(16,17)18)7(19,20)21)30-3(8(22,23)24)9(25,26)27/h1-3H
InChI 密鑰
MJOVEPJSFHDSOJ-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
Tris(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite (tris(1,1,1,3,3,3-hexafluoroisopropyl) phosphite) is a sterically hindered, weakly σ-donating and strongly π-accepting ligand. It can be prepared by the reaction of lithium salt of 1,1,1,3,3,3-hexafluoro-2-propoxide with PCl3. It reacts with nucleosides to form deoxyribonucleoside 3′-bis(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite units.
應用
Tris(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite (tris(1,1,1,3,3,3-hexafluoroisopropyl) phosphite) may be used in the following processes:
- Preparation of new phosphonylating and coupling agents for the synthesis of oligodeoxyribonucleotides.
- Preparation of nucleoside H-phosphonate units.
- Synthesis of tetrakis(1,1,1,3,3,3-hexafluoroisopropoxy)(phenylthio)phosphorane by reacting with 1,1,1,3,3,3-hexafluoroisopropyl benzenesulfenate.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
Preparation and nuclear magnetic resonance studies of pentacoordinated phosphorus compounds containing hexafluoroisopropoxy groups.
Denney DB, et al.
The Journal of Organic Chemistry, 48(13), 2159-2164 (1983)
Stereoselective propargylation mediated by a chiral metal cluster: reactions of [(propargylium)Co2 (CO)5{P(OR)3}][BF4] with carbon nucleophiles.
Caffyn AJM and Nicholas KM.
Journal of the American Chemical Society, 115(14), 6438-6439 (1993)
O Sakatsume et al.
Nucleic acids research, 18(11), 3327-3331 (1990-06-11)
New phosphonylating and coupling agents for the synthesis of oligodeoxyribonucleotides via H-phosphonate approach have been developed. Tris(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite, prepared by the reaction of lithium salt of 1,1,1,3,3,3-hexafluoro-2-propoxide with PCl3, reacts with deoxyribonucleosides in the presence of a catalytic amount of
Synthesis of antisense oligodeoxyribonucleotide analogues by use of deoxyribonucleoside 3'-bis(1,1, 1,3,3,3-hexafluoro-2-propyl)phosphites as new key intermediates.
Hosaka H, et al.
Heteroatom Chem., 2(1), 197-204 (1991)
O Sakatsume et al.
Nucleic acids research, 17(10), 3689-3697 (1989-05-25)
The solid phase synthesis of oligoribonucleotides using the H-phosphonate approach and the 1-[(2-chloro-4-methyl)phenyl]-4-methoxypiperidin-4-yl (Ctmp) and dimethoxytrityl (DMTr) groups, respectively, for the protection of the 2'- and 5'-hydroxy functions is described. The use of a new reagent, tris-(1,1,1,3,3,3-hexafluoro-2-propyl) phosphite for the
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務



![4-叔丁基杯[4]芳烃 95%](/deepweb/assets/sigmaaldrich/product/structures/141/993/6110bb7e-274f-45f8-887a-f1841a7dae7c/640/6110bb7e-274f-45f8-887a-f1841a7dae7c.png)