全部照片(1)
About This Item
經驗公式(希爾表示法):
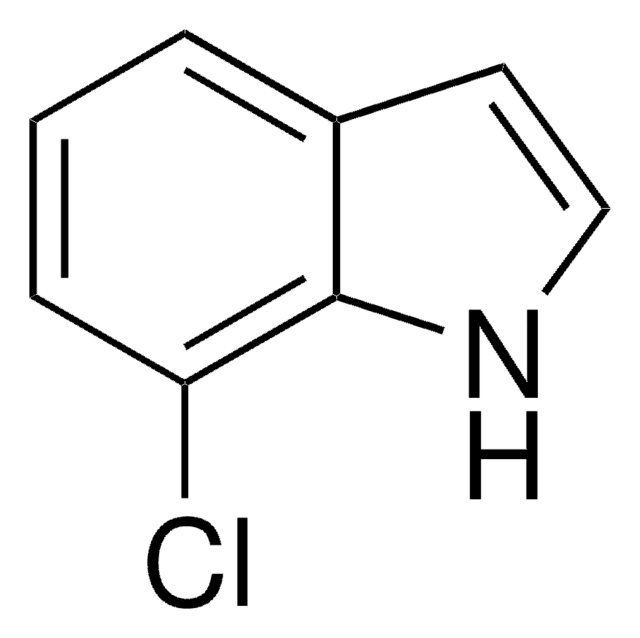
C8H6ClN
CAS號碼:
分子量::
151.59
Beilstein:
114880
EC號碼:
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
暫時無法取得訂價和供貨情況
推薦產品
化驗
98%
折射率
n20/D 1.628 (lit.)
bp
129-130 °C/4 mmHg (lit.)
溶解度
ethanol: 50 mg/mL, clear, colorless
密度
1.259 g/mL at 25 °C (lit.)
官能基
chloro
SMILES 字串
Clc1cccc2[nH]ccc12
InChI
1S/C8H6ClN/c9-7-2-1-3-8-6(7)4-5-10-8/h1-5,10H
InChI 密鑰
SVLZRCRXNHITBY-UHFFFAOYSA-N
尋找類似的產品? 前往 產品比較指南
一般說明
The nitrosation rate of 4-chloroindole and the stability of its nitrosated products were studied[1].
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
235.4 °F - closed cup
閃點(°C)
113 °C - closed cup
個人防護裝備
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
客戶也查看了
H G Tiedink et al.
Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 27(11), 723-730 (1989-11-01)
The nitrosation rates of indole-3-acetonitrile, indole-3-carbinol, indole and 4-chloroindole and the stability of their nitrosated products were investigated. Each of the nitrosated indole compounds was directly mutagenic to Salmonella typhimurium TA100 in the following order of potency: 4-chloroindole much greater
Zhong-Liu Wu et al.
The Journal of biological chemistry, 280(49), 41090-41100 (2005-10-11)
The natural product indole is a substrate for cytochrome P450 2A6. Mutagenesis of P450 2A6 was done to expand its capability in the oxidization of bulky substituted indole compounds, which are not substrates for the wild-type enzyme or the double
Zhi-Gang Zhang et al.
Journal of biotechnology, 139(1), 12-18 (2008-11-06)
Cytochrome P450 (P450) 2A6 is able to catalyze indole hydroxylation to form the blue dye indigo. The wild-type P450 2A6 enzyme was randomly mutated throughout the whole open reading frame and screened using 4-chloroindole hydroxylation, a substituted indole selected from
H G Tiedink et al.
Cell biology and toxicology, 7(4), 371-386 (1991-10-01)
4-chloro-methoxyindole is a naturally occurring compound in Vicia faba which can easily react with nitrite to form a N-nitroso compound. In this in vitro study, the potential genotoxic effects of nitrosated 4-chloro-6-methoxyindole and its structural analogue 4-chloroindole were evaluated for
Chaitany Jayprakash Raorane et al.
Biomolecules, 10(8) (2020-08-23)
Multi-drug resistant Acinetobacter baumannii is well-known for its rapid acclimatization in hospital environments. The ability of the bacterium to endure desiccation and starvation on dry surfaces for up to a month results in outbreaks of health care-associated infections. Previously, indole
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務