推薦產品
等級
technical grade
品質等級
蒸汽壓力
10 mmHg ( 20 °C)
化驗
85%
形狀
liquid
折射率
n20/D 1.488 (lit.)
bp
74-76 °C/40 mmHg (lit.)
mp
1-3 °C (lit.)
密度
1.183 g/mL at 25 °C (lit.)
官能基
chloro
儲存溫度
2-8°C
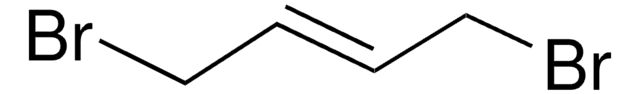
SMILES 字串
ClC\C=C\CCl
InChI
1S/C4H6Cl2/c5-3-1-2-4-6/h1-2H,3-4H2/b2-1+
InChI 密鑰
FQDIANVAWVHZIR-OWOJBTEDSA-N
尋找類似的產品? 前往 產品比較指南
訊號詞
Danger
危險分類
Acute Tox. 1 Inhalation - Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1B
儲存類別代碼
3 - Flammable liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
127.4 °F - closed cup
閃點(°C)
53 °C - closed cup
個人防護裝備
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Z M Wang et al.
Chirality, 12(7), 581-589 (2000-06-22)
A highly efficient synthetic method for the trans-tetrahydrofuran (THF) ring building block was established and the title compound was synthesized in 19 steps from trans-1,4-dichloro-2-butene via a convergent route with a Wittig reaction as the key step.
A D Elbein et al.
Biochemistry, 26(9), 2502-2510 (1987-05-05)
The chemical synthesis of swainsonine [(1S,2R,8R,8 alpha R)-trihydroxyindolizidine] from trans-1,4-dichloro-2-butene was previously described [Adams, C. E., Walker, F. J., & Sharpless, K. B. (1985) J. Org. Chem. 50, 420-424]. A modification of that synthesis provided two other isomers, referred to
S Phadtare et al.
Journal of medicinal chemistry, 30(2), 437-440 (1987-02-01)
Alkylation of adenine (5a) or 2-amino-6-chloropurine (5b) with excess trans-1,4-dichloro-2-butene (4), effected by K2CO3 in dimethyl sulfoxide or tetra-n-butylammonium fluoride in tetrahydrofuran, led in 90-95% regioselectivity to 9-alkylpurines 6a and 6b. The title compounds 2a and 2b were obtained by
[Experimental data on a hygienic standard for 1,4-dichlorobutene-2 in the air of a work area].
M S Gizhlarian et al.
Gigiena truda i professional'nye zabolevaniia, (4)(4), 49-50 (1985-04-01)
S Phadtare et al.
Nucleic acids symposium series, (18)(18), 25-28 (1987-01-01)
Reaction of adenine (1a) or cytosine (1b) with excess 1,4-dichloro-2-butyne catalyzed by K2CO3 in (CH3)2SO gave the 4-chloro-2-butynyl derivatives 2a and 2b. The latter were converted to the 4-hydroxy-2-butynyl compounds 3a and 3b by refluxing in 0.1 M HCl. Isomerization
Active Filters
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務