所有图片(1)
About This Item
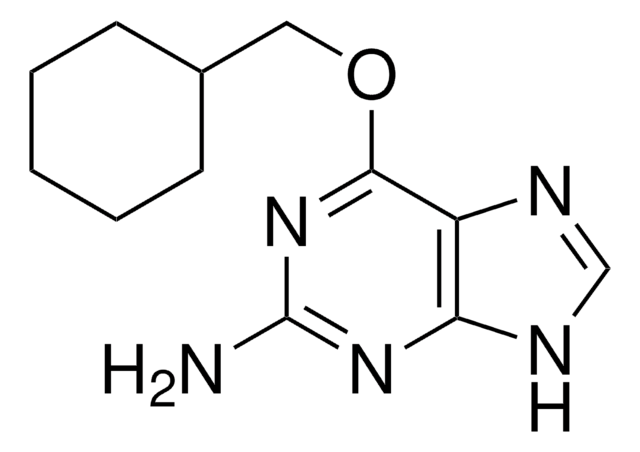
经验公式(希尔记法):
C16H17N2O5SK
分子量:
388.48
EC號碼:
MDL號碼:
分類程式碼代碼:
12352106
PubChem物質ID:
NACRES:
NA.85
推荐产品
形狀
solid
效力
1444-1536 units per mg
抗生素活性譜
Gram-positive bacteria
作用方式
cell wall synthesis | interferes
SMILES 字串
[K+].CC1(C)S[C@@H]2[C@H](NC(=O)COc3ccccc3)C(=O)N2[C@H]1C([O-])=O
InChI
1S/C16H18N2O5S.K/c1-16(2)12(15(21)22)18-13(20)11(14(18)24-16)17-10(19)8-23-9-6-4-3-5-7-9;/h3-7,11-12,14H,8H2,1-2H3,(H,17,19)(H,21,22);/q;+1/p-1/t11-,12+,14-;/m1./s1
InChI 密鑰
HCTVWSOKIJULET-LQDWTQKMSA-M
正在寻找类似产品? 访问 产品对比指南
一般說明
Chemical structure: ß-lactam
應用
Phenoxymethylpenicillinic acid, also known as Penicillin V, is a narrow spectrum antibiotic used to treat mild to moderate infections caused by susceptible bacteria. It is a natural penicillin antibiotic that is administered orally. Penicillin V is a penicillin β-lactam antibiotic used in the treatment of bacterial infections caused by susceptible, usually gram-positive, organisms. It is commonly used in cell culture in combination with streptomcyin and other antibiotics. It is used to study penicillin-binding protein 2, molecular cloning of the Bacillus sphaericus penicillin V amidase gene, and gastrointestinal persistence and bile tolerance of Listeria monocytogenes.
生化/生理作用
Phenoxymethylpenicillinic acid binds to specific penicillin-binding proteins (PBPs) and inhibits the synthesis of bacterial cell walls by preventing cell wall peptidoglycan chain cross-linking. This causes cell lysis, which is mediated by autolytic enzymes such as autolysins. Phenoxymethylpenicillnic acid may interfere with autolysin inhibitors.
其他說明
10mu,100mu
Keep container tightly closed in a dry and well-ventilated place.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Resp. Sens. 1 - Skin Sens. 1
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
A Olsson et al.
Applied and environmental microbiology, 49(5), 1084-1089 (1985-05-01)
The Bacillus sphaericus gene coding for penicillin V amidase, which catalyzes the hydrolysis of penicillin V to yield 6-aminopenicillanic acid and phenoxyacetic acid, has been isolated by molecular cloning in Escherichia coli. The gene is contained within a 2.2-kilobase HindIII-PstI
Máire Begley et al.
Infection and immunity, 73(2), 894-904 (2005-01-25)
Listeria monocytogenes must resist the deleterious actions of bile in order to infect and subsequently colonize the human gastrointestinal tract. The molecular mechanisms used by the bacterium to resist bile and the influence of bile on pathogenesis are as yet
Susumu Ochiai et al.
The Journal of antimicrobial chemotherapy, 60(1), 54-60 (2007-06-02)
In Neisseria gonorrhoeae, the mosaic structure of penicillin-binding protein 2 (PBP 2), composed of fragments of PBP 2 from Neisseria cinerea and Neisseria perflava, was significantly associated with decreased susceptibility to cephalosporins, particularly oral cephalosporins. The aim of this study
Adel Ben Salem et al.
Bioorganic & medicinal chemistry, 19(24), 7534-7540 (2011-11-15)
Two well-known antibiotic heterocycles, the 'quinolone' nalidixic acid and the β-lactam penicillin V, active at different levels of the bacterial growth process, have been attached via an ether-ester junction to the p-tert-butylcalix[4]arene lower rim, in alternate position. The resulting hydrophobic
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门