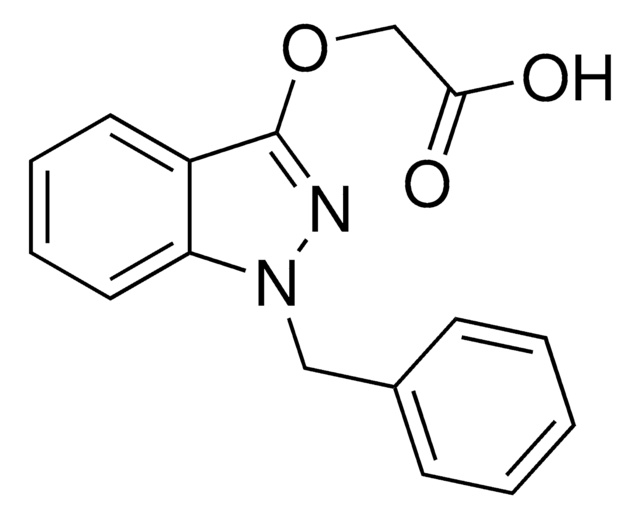
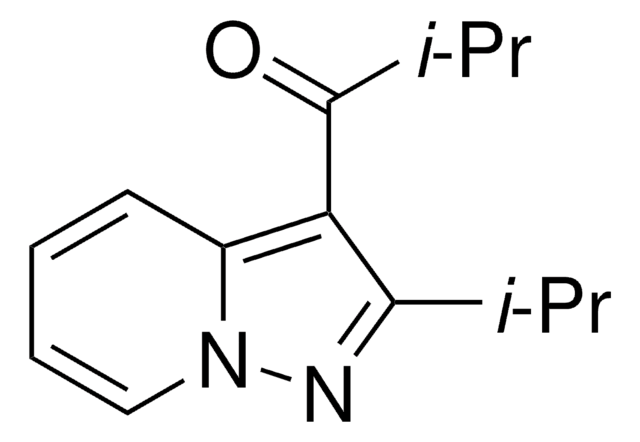
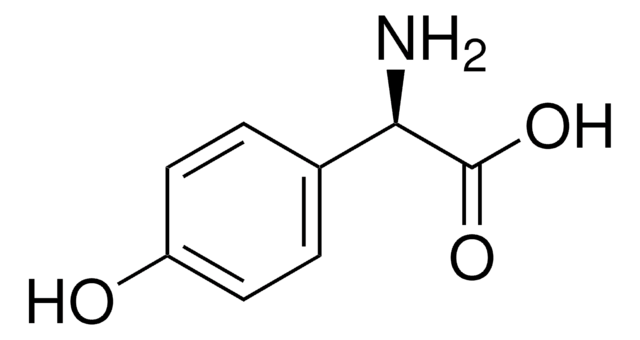
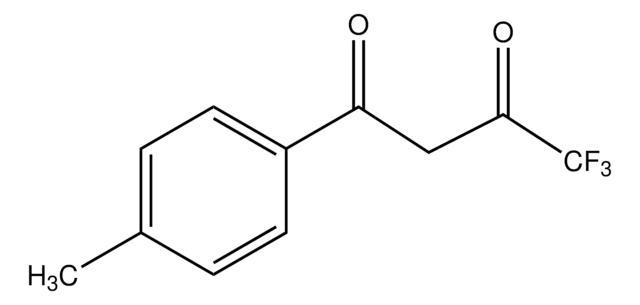
推荐产品
等级
pharmaceutical primary standard
API类
emedastine
制造商/商品名称
EDQM
应用
pharmaceutical (small molecule)
包装形式
neat
储存温度
2-8°C
SMILES字符串
N1(CCN(CCC1)c2[n](c3c(n2)cccc3)CCOCC)C.OC(=O)\C=C\C(=O)O.OC(=O)\C=C\C(=O)O
InChI
1S/C17H26N4O.2C4H4O4/c1-3-22-14-13-21-16-8-5-4-7-15(16)18-17(21)20-10-6-9-19(2)11-12-20;2*5-3(6)1-2-4(7)8/h4-5,7-8H,3,6,9-14H2,1-2H3;2*1-2H,(H,5,6)(H,7,8)/b;2*2-1+
InChI key
FWLKKPKZQYVAFR-LVEZLNDCSA-N
正在寻找类似产品? 访问 产品对比指南
相关类别
一般描述
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
应用
Emedastine difumarate EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他说明
Sales restrictions may apply.
警示用语:
Warning
危险声明
危险分类
Acute Tox. 4 Oral
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
Yuan Tian et al.
Journal of chromatographic science, 45(3), 158-164 (2007-04-28)
A selective and sensitive method employing high-performance liquid chromatography (HPLC)-electrospray ionization (ESI)-mass spectrometry is developed and validated for the determination of emedastine difumarate in human plasma. With naphazoline hydrochloride as the internal standard, emedastine difumarate is extracted from plasma with
Mario E Corrado et al.
Arzneimittel-Forschung, 54(10), 660-665 (2004-11-24)
Emedastine is a new H1-receptor antagonist endowed with potent and selective antihistamine activity. The aim of this study was to evaluate the therapeutic efficacy and tolerability of emedastine difumarate (CAS 87233-62-3) in Caucasian patients in the treatment of seasonal allergic
Shoichi Harada et al.
Journal of pharmaceutical sciences, 94(1), 17-24 (2005-03-12)
Antihistamine effects of emedastine applied topically with three vehicles varying in their polarities were investigated in rats. The pharmacological effect of emedastine differed greatly depending on its concentration, treatment time, and vehicle. The antihistamine effect reached a plateau after approximately
László Endre
Orvosi hetilap, 148(6), 251-254 (2007-03-09)
In the treatment of any disease, it is advantageous if the concentration of the administered drug is highest in the target organ. It follows that, if possible, diseases of the conjunctiva should primarily be treated with eye drops. The author
Annik Pons-Guiraud et al.
European journal of dermatology : EJD, 16(6), 649-654 (2007-01-19)
Emedastine difumarate (2 mg b.i.d.) was compared to loratadine (10 mg o.d.) in a randomized, double-blind, multicentre trial for 4 weeks in 192 patients with idiopathic chronic urticaria. After one week of treatment significant differences were recorded: body skin involvement
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门