所有图片(1)
选择尺寸
变更视图
50 G
$54.50
250 G
$166.00
1 KG
$370.00
About This Item
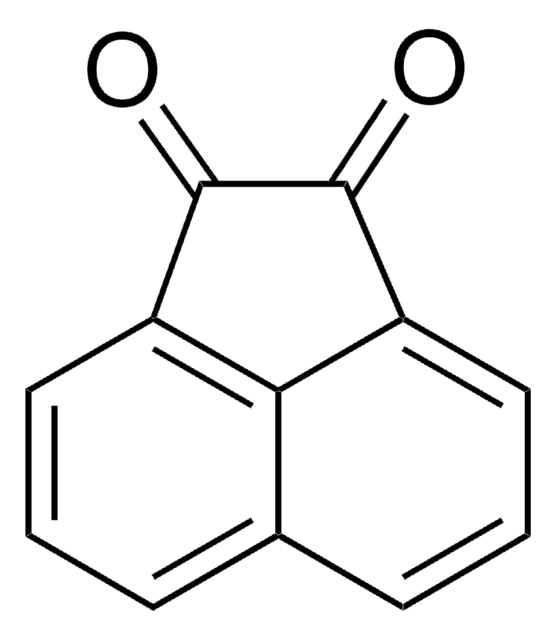
经验公式(希尔记法):
C10H6O2
CAS号:
分子量:
158.15
Beilstein:
878524
EC 号:
MDL编号:
UNSPSC代码:
12352100
PubChem化学物质编号:
NACRES:
NA.22
推荐产品
等级
purum
质量水平
方案
≥96.5% (HPLC)
表单
powder
mp
119-122 °C (lit.)
120-124 °C
官能团
ketone
SMILES字符串
O=C1C=CC(=O)c2ccccc12
InChI
1S/C10H6O2/c11-9-5-6-10(12)8-4-2-1-3-7(8)9/h1-6H
InChI key
FRASJONUBLZVQX-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
应用
1,4-Naphthoquinone is the key structural moiety of many anticancer and antifungal agents.[1]
It can be used to synthesize:[2]
Additional appilcation include:[2]
It can be used to synthesize:[2]
- 3,3-Disubstituted oxindoles via asymmetric Michael addition to oxindole.
- Bioactive isoindolines via asymmetric 1,3-dipolar cycloaddition to azomethine ylides generated in situ from aldehydes and diethyl aminomalonate.
- α,α-Difluoro-β-hydroxy ketone via ‘on water′ catalyst-free Mukaiyama-aldol reaction with difluoroenoxysilane.
- 2-Hydroxy-3-anilino-1,4-naphthoquinone, which shows potent in vivo antimalarial activity.
Additional appilcation include:[2]
- As an arylation reagent for the α-arylation of aldehydes.
- As a starting material in the multi-step synthesis of benz[f]indole-4,9-diones.
警示用语:
Danger
危险分类
Acute Tox. 1 Inhalation - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1C - Skin Sens. 1 - STOT SE 3
靶器官
Respiratory system
储存分类代码
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
闪点(°F)
285.8 °F
闪点(°C)
141 °C
个人防护装备
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
其他客户在看
Design, synthesis and evaluation of novel 1, 4-naphthoquinone derivatives as antifungal and anticancer agents.
Tandon V K, et al.
Bioorganic & Medicinal Chemistry Letters, 14(5), 1079-1083 (2004)
1, 4-Naphthoquinone.
Yu J S.
Synlett, 25(16), 2377-2378 (2014)
Jan Bitenc et al.
Materials (Basel, Switzerland), 13(3) (2020-01-25)
Organic cathode materials are promising cathode materials for multivalent batteries. Among organic cathodes, anthraquinone (AQ) has already been applied to various metal‒organic systems. In this work, we compare electrochemical performance and redox potential of AQ with 1,4-naphthoquinone (NQ) and 1,4-benzoquinone
Don Antoine Lanfranchi et al.
Organic & biomolecular chemistry, 10(31), 6375-6387 (2012-07-11)
Improving the solubility of polysubstituted 1,4-naphthoquinone derivatives was achieved by introducing nitrogen in two different positions of the naphthoquinone core, at C-5 and at C-8 of menadione through a two-step, straightforward synthesis based on the regioselective hetero-Diels-Alder reaction. The antimalarial
Michael G Berg et al.
Molecular and cellular biology, 32(7), 1271-1283 (2012-01-19)
Despite intensive research, there are very few reagents with which to modulate and dissect the mRNA splicing pathway. Here, we describe a novel approach to identify such tools, based on detection of the exon junction complex (EJC), a unique molecular
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门