推荐产品
方案
99%
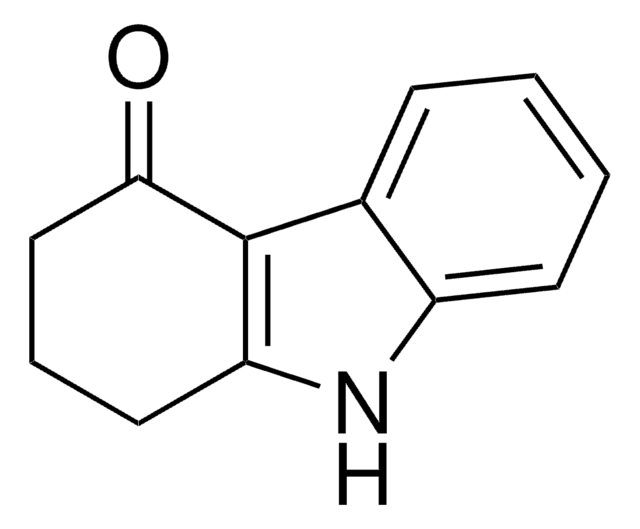
沸点
325-330 °C (lit.)
mp
118-120 °C (lit.)
SMILES字符串
C1CCc2c(C1)[nH]c3ccccc23
InChI
1S/C12H13N/c1-3-7-11-9(5-1)10-6-2-4-8-12(10)13-11/h1,3,5,7,13H,2,4,6,8H2
InChI key
XKLNOVWDVMWTOB-UHFFFAOYSA-N
应用
1,2,3,4-四氢咔唑可作为起始材料:
- 通过光氧化反应制备螺[环戊烷-1,2′-吲哚啉-3′-酮]。
- 通过N-酰基化反应制备9-酰基-1,2,3,4-四氢咔唑。
- 通过钯催化的不对称加氢反应制备咔唑。
警示用语:
Warning
危险声明
危险分类
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
靶器官
Respiratory system
储存分类代码
11 - Combustible Solids
WGK
WGK 2
闪点(°F)
Not applicable
闪点(°C)
Not applicable
个人防护装备
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
Romano Di Fabio et al.
Bioorganic & medicinal chemistry letters, 16(6), 1749-1752 (2005-12-21)
The SAR of a new series of tetrahydrocarbazole derivatives is described: the appropriate decoration of this template led to the identification of a new class of NPY-1 antagonists showing good in vitro potency and a promising in vivo pharmacokinetic profile
Subhasish Neogi et al.
Journal of combinatorial chemistry, 12(5), 617-629 (2010-07-01)
The one-pot synthesis of a new substituted 1,2,3,4-tetrahydrocarbazoles has been described via Petasis reactions. These tetrahydrocarbazoles exhibits various medicinal importance and might be suitable for elaboration into larger peptides at carboxy termini. The scope and limitations of this method have
J Cao et al.
Chemosphere, 40(12), 1411-1416 (2000-05-02)
The solubilization of four pairs of substituted indole compounds (SICs) by beta-cyclodextrin (beta-CD) in water was investigated. The results show that 1,2,3,4-tetrahydrocarbazole and N-methyl-1,2,3,4-tetrahydrocarbazole form 1:1 inclusion complexes with beta-CD, while the other six SICs form 1:2 inclusion complexes, respectively.
Chao Zheng et al.
Accounts of chemical research, 53(4), 974-987 (2020-04-11)
ConspectusThe Pictet-Spengler reaction is a fundamental named reaction in organic chemistry, and it is the most straightforward method for the synthesis of tetrahydro-β-carbolines, a core structure embedded in numerous alkaloids. Spiroindolenines are often proposed as possible intermediates in Pictet-Spengler reactions.
Jean Chang-Fong et al.
Bioorganic & medicinal chemistry letters, 14(8), 1961-1964 (2004-03-31)
An investigation of the structure-affinity relationships for the binding of 4-(N,N-dimethylaminomethyl)-N(9)-arylsulfonyl-9H-1,2,3,4-tetrahydrocarbazoles (conformationally-constrained analogues of the benzenesulfonyltryptamine 5-HT(6) antagonist MS-245) at human 5-HT(6) receptors revealed that various arylsulfonyl substituents are tolerated and that the 4-(N,N-dimethylaminomethyl) group is not required for binding.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门