推荐产品
化驗
98%
形狀
liquid
光學活性
[α]20/D +54°, neat
折射率
n20/D 1.439 (lit.)
bp
80-82 °C/0.1 mmHg (lit.)
密度
1.19 g/mL at 25 °C (lit.)
SMILES 字串
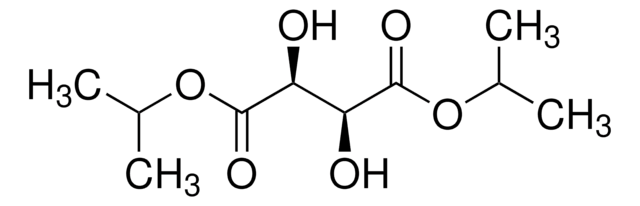
COC(=O)[C@H]1OC(C)(C)O[C@@H]1C(=O)OC
InChI
1S/C9H14O6/c1-9(2)14-5(7(10)12-3)6(15-9)8(11)13-4/h5-6H,1-4H3/t5-,6-/m0/s1
InChI 密鑰
ROZOUYVVWUTPNG-WDSKDSINSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
(+)-Dimethyl 2,3-O-isopropylidene-D-tartrate is a chiral reagent used in organic synthesis. It is prepared by the reaction-rectification process from D -tartrate, and 2,2-dimethoxypropane.{138]
應用
TADDOL 手性助剂、联吡啶配体和苏糖醇的有用结构单元。
(+)-Dimethyl 2,3-O-isopropylidene-D-tartrate can be used as a starting material for the synthesis of:
- Deuterated 1-deoxy-D-xylose.
- C-terminal peptide -oxo-aldehydes using Fmoc solid-phase peptide synthesis methodology (SPPS).
- Bis-Weinreb amide, a key intermediate for the preparation of myo-inositol analog via ring-closing metathesis.
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
275.0 °F - closed cup
閃點(°C)
135 °C - closed cup
個人防護裝備
Eyeshields, Gloves
A new linker for the synthesis of C-terminal peptide ?-oxo-aldehydes
Fruchart J-S, et al.
Tetrahedron Letters, 40(34), 6225-6228 (1999)
Rosemary M Conrad et al.
Organic letters, 4(8), 1359-1361 (2002-04-13)
Here we report a concise stereoselective synthesis of myo-inositol via ring-closing metathesis. A readily available bis-Weinreb amide of D-tartrate served as a key intermediate. [reaction: see text]
Angewandte Chemie (International Edition in English), 30, 99-99 (1991)
Organic Syntheses, 68, 92-92 (1990)
The Journal of Organic Chemistry, 57, 6825-6825 (1992)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门