推荐产品
化驗
98%
mp
80-82 °C (lit.)
SMILES 字串
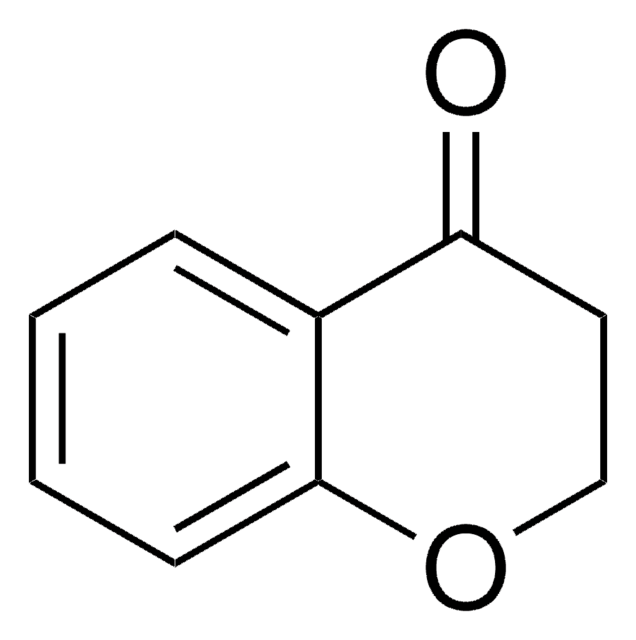
O=C1Cc2ccccc2CO1
InChI
1S/C9H8O2/c10-9-5-7-3-1-2-4-8(7)6-11-9/h1-4H,5-6H2
InChI 密鑰
ILHLUZUMRJQEAH-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
3-Isochromanone has been reported to be isolated from the fungus Nigrospora sp. PSU-F12. An improved Knoevenagel condensation of 3-isochromanone with aromatic aldehydes has been achieved by microwave irradiation on solid supports in the presence of various catalysts. Synthesis of 3-isochromanone via Beayer-Villiger rearrangement has been reported.
應用
3-Isochromanone may be used as starting reagent in the synthesis of of BDPBI (7-bromo-1,4-dihydro-2-phenyl-4,4-bis(4-pyridinylmethyl)2H-isoquinolin-3-one dihydrochloride).
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
András Vass et al.
Journal of biochemical and biophysical methods, 69(1-2), 179-187 (2006-05-09)
An improved Knoevenagel condensation of 3-isochromanone and aromatic aldehydes can be achieved by microwave irradiation on solid supports in the presence of various catalysts. This synthetic method offers some major advantages, especially the possibility to change the ratio of E/Z
Chia Hsien Lin et al.
Pharmacology, 67(4), 202-210 (2003-02-22)
The pharmacological effects of BDPBI (7-bromo-1,4-dihydro-2-phenyl-4,4-bis(4-pyridinylmethyl)2H-isoquinolin-3-one dihydrochloride) were tested on isolated endothelium-containing or denuded aorta of the guinea pig. BDPBI with the formula C(27)H(24)BrCl(2)N(3)O was synthesized starting with 3-isochromanone. In the endothelium-containing preparations of the aortic rings, phenylephrine (PHE; 10
Vatcharin Rukachaisirikul et al.
Archives of pharmacal research, 33(3), 375-380 (2010-04-03)
Nigrosporanenes A (1) and B (2), two new cylohexene derivatives, and tyrosol (3) were isolated from the sea fan-derived fungus Nigrospora sp. PSU-F11, whereas five known compounds: 4-hydroxybenzoic acid (4), aplysiopsene D (5), 3-isochromanone (6), (-)-drimenin (7) and diketopiperazine derivative
3-Isochromanone.
Markgrar JH and Basta SJ.
Synthetic Communications, 2(3), 139-141 (1972)
商品
Knoevenagel Condensation is an organic reaction named after Emil Knoevenagel. It is a classic C-C bond formation reaction and a modification of the Aldol Condensation.
相关内容
Knoevenagel Condensation is an organic reaction named after Emil Knoevenagel. It is a classic C-C bond formation reaction and a modification of the Aldol Condensation.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门