Zaloguj się, aby wyświetlić ceny organizacyjne i kontraktowe.
Wybierz wielkość
100 MG
556,00 zł
556,00 zł
Skontaktuj się z Obsługą Klienta, aby uzyskać informacje na temat dostępności
Informacje o tej pozycji
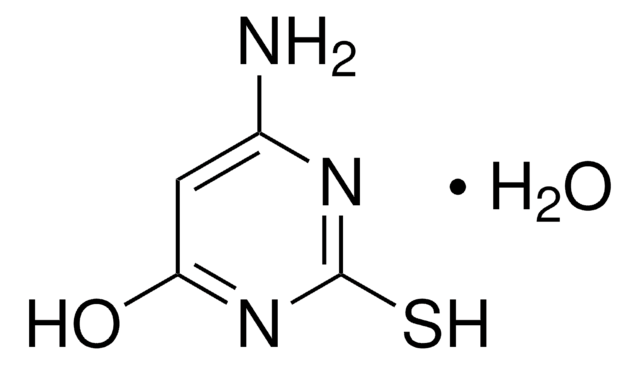
Wzór empiryczny (zapis Hilla):
C6H7N5S
Numer CAS:
Masa cząsteczkowa:
181.22
NACRES:
NA.51
PubChem Substance ID:
UNSPSC Code:
41106305
EC Number:
214-833-4
MDL number:
Assay:
≥95%
Form:
solid
Przejdź do
Pomoc techniczna
Potrzebujesz pomocy? Nasz zespół doświadczonych naukowców chętnie Ci pomoże.
Pozwól nam pomócQuality Level
assay
≥95%
form
solid
SMILES string
[H]n1cnc2c(SC)nc(N)nc12
InChI
1S/C6H7N5S/c1-12-5-3-4(9-2-8-3)10-6(7)11-5/h2H,1H3,(H3,7,8,9,10,11)
InChI key
YEGKYFQLKYGHAR-UHFFFAOYSA-N
1 of 4
Ta pozycja | |||
|---|---|---|---|
| Quality Level 200 | Quality Level 200 | Quality Level 100 | Quality Level 100 |
| assay ≥95% | assay 97% | assay 98% | assay ≥96.5% (HPLC), 97% |
| form solid | form solid | form crystals | form crystals |
Application
2-Amino-6-methylmercaptopurine is a 2-amino-6-alkyldithiopurine that has been used with other 6-position carbon analogues to study brain specific diazepam binding.
2-Amino-6-methylmercaptopurine (6-MTG) has been used as a supplement in Dulbecco′s modified Eagles medium (DMEM) medium for the selection of gpt-expressing recombinant virus mCMVhMIEPE-gpt.lacZ (cytomegalovirus major immediate-early promotor-enhancer complex-gpt.lacz). It has also been used as a standard in high performance liquid chromatography (HPLC) to assess the activity of thiopurine methyltransferase (TPMT) enzyme.
Biochem/physiol Actions
2-Amino-6-methylmercaptopurine is synthesized from 6-mercaptopurine by the S methylation activity of thiopurine methyl transferase (TMPT) enzyme.
Ta strona może zawierać tekst przetłumaczony maszynowo.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Klasa składowania
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
S C Sung et al.
Biochemical pharmacology, 33(11), 1737-1739 (1984-06-01)
Various derivatives of 2-amino-6- methylthiopurine with substituents at the 6-position of purine were tested for their abilities to displace [3H]diazepam binding to rat brain membranes. The potency was dependent on the carbon chain-length in the 6-position of purine. Among the
Effect of 9-alkyl derivatives of 6-methylthioguanine on brain specific binding of [3H]diazepam.
S C Sung et al.
Biochemical pharmacology, 35(20), 3645-3646 (1986-10-15)
A Practical Non-Extraction Direct Liquid Chromatography Method for Determination of Thiopurine S-Methyltransferase Activity in Inflammatory Bowel Disease
Bahrehmand F, et al.
Acta Medica Iranica, 55(6), 360-367 (2017)
G R Erdmann et al.
Biomedical chromatography : BMC, 4(2), 47-51 (1990-03-01)
A reversed phase high performance liquid chromatographic procedure was developed to quantify 6-thioguanine, 6-mercaptopurine, methylthioguanine, and methylmercaptopurine in red blood cells. The free base of each thiopurine was liberated from the respective nucleoside and nucleotide moiety by acid hydrolysis, which
S C Sung et al.
European journal of pharmacology, 81(3), 505-508 (1982-07-16)
We have compared fifteen synthetic purines and purine nucleosides on their ability to displace [3H]diazepam binding to rat brain membranes. Among these analogs, 6-methylthioguanine was found to be most potent, inhibiting competitively the specific binding of [3H]diazepam with a Ki
Numer pozycji handlu globalnego
| SKU | NUMER GTIN |
|---|---|
| A9546-100MG | 04061833045381 |
Active Filters
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej