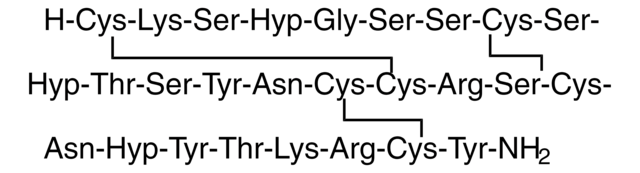About This Item
推荐产品
质量水平
方案
≥97% (HPLC)
储存温度
−20°C
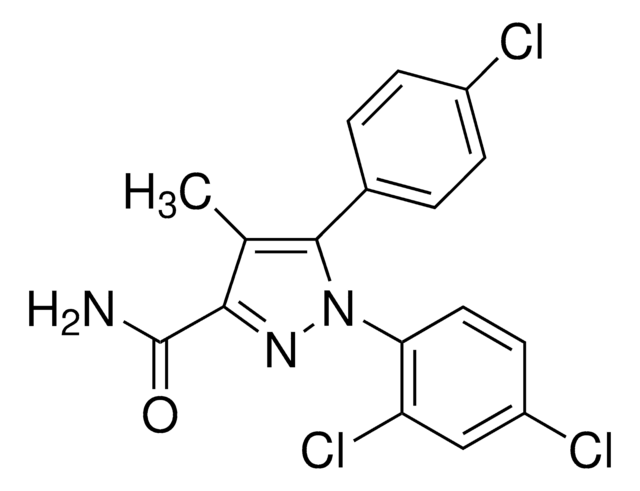
SMILES字符串
CCC(C)C(NC(=O)C(NC(=O)C(CC(O)=O)NC(=O)C(CCC(N)=O)NC(=O)C(Cc1cnc[nH]1)NC(=O)C2CSSCC(N)C(=O)NC(C(C)O)C(=O)NC3CSSCC(NC(=O)C(CCC(O)=O)NC(=O)C(CCC(O)=O)NC(=O)C(CC(O)=O)NC(=O)C(NC(=O)C(CCSC)NC(=O)C(CC(O)=O)NC(=O)C(CC(N)=O)NC3=O)C(C)O)C(=O)NC(CC(C)C)C(=O)NC(CC(N)=O)C(=O)NC(Cc4ccccc4)C(=O)N2)C(C)C)C(=O)NC(Cc5c[nH]c6ccccc56)C(O)=O
InChI
1S/C103H147N27O37S5/c1-10-46(6)80(100(163)123-67(103(166)167)30-50-37-109-54-19-15-14-18-52(50)54)128-99(162)79(45(4)5)127-95(158)66(36-78(144)145)120-85(148)55(20-23-71(105)133)112-90(153)61(31-51-38-108-43-110-51)117-97(160)69-40-170-169-39-53(104)83(146)129-81(47(7)131)102(165)126-70-42-172-171-41-68(96(159)115-59(28-44(2)3)88(151)118-62(32-72(106)134)91(154)116-60(89(152)125-69)29-49-16-12-11-13-17-49)124-86(149)57(22-25-75(138)139)111-84(147)56(21-24-74(136)137)113-94(157)65(35-77(142)143)122-101(164)82(48(8)132)130-87(150)58(26-27-168-9)114-93(156)64(34-76(140)141)121-92(155)63(33-73(107)135)119-98(70)161/h11-19,37-38,43-48,53,55-70,79-82,109,131-132H,10,20-36,39-42,104H2,1-9H3,(H2,105,133)(H2,106,134)(H2,107,135)(H,108,110)(H,111,147)(H,112,153)(H,113,157)(H,114,156)(H,115,159)(H,116,154)(H,117,160)(H,118,151)(H,119,161)(H,120,148)(H,121,155)(H,122,164)(H,123,163)(H,124,149)(H,125,152)(H,126,165)(H,127,158)(H,128,162)(H,129,146)(H,130,150)(H,136,137)(H,138,139)(H,140,141)(H,142,143)(H,144,145)(H,166,167)
InChI key
LXPHPKVWHQLBBA-UHFFFAOYSA-N
基因信息
human ... EDNRB(1910)
mouse ... EDNRB(13618)
rat ... EDNRB(50672)
正在寻找类似产品? 访问 产品对比指南
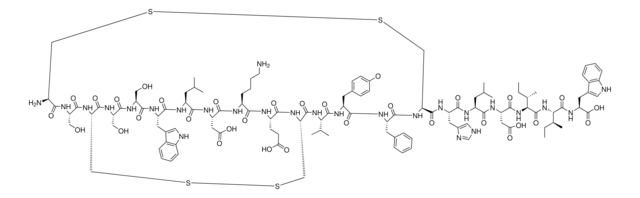
Amino Acid Sequence
生化/生理作用
其他说明
警示用语:
Danger
危险声明
危险分类
Acute Tox. 1 Inhalation - Acute Tox. 1 Oral
储存分类代码
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
个人防护装备
Eyeshields, Gloves, type N95 (US)
历史批次信息供参考:
分析证书(COA)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门