This product is reasonably priced compared to other suppliers.
F5130
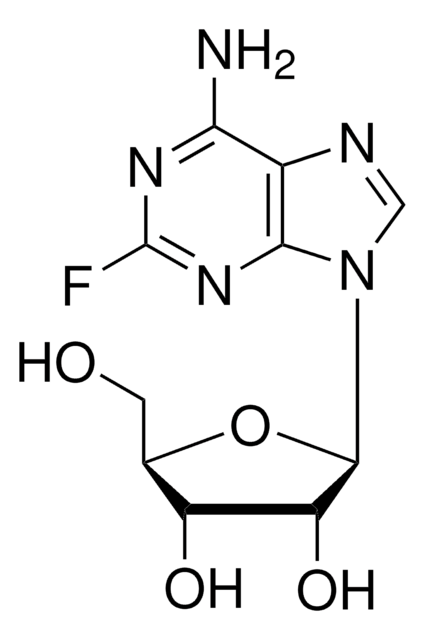
5-氟脲嘧啶
5-Fluorouridine
1 of 1 reviewers received a sample product or took part in a promotion
proapoptotic anitproliferative plant growth regulator
别名:
5-氟尿嘧啶 1β-D-呋喃核苷, 皮毛
登录查看公司和协议定价
所有图片(4)
About This Item
经验公式(希尔记法):
C9H11FN2O6
CAS号:
分子量:
262.19
Beilstein:
33662
EC 号:
MDL编号:
UNSPSC代码:
41106305
PubChem化学物质编号:
NACRES:
NA.51
价格与库存信息目前不能提供
推荐产品
生物来源
synthetic (organic)
方案
≥99% (HPLC)
表单
powder
溶解性
water: 50 mg/mL, clear to slightly hazy, colorless to faintly yellow
SMILES字符串
OC[C@H]1O[C@H]([C@H](O)[C@@H]1O)N2C=C(F)C(=O)NC2=O
InChI
1S/C9H11FN2O6/c10-3-1-12(9(17)11-7(3)16)8-6(15)5(14)4(2-13)18-8/h1,4-6,8,13-15H,2H2,(H,11,16,17)/t4-,5-,6-,8-/m1/s1
InChI key
FHIDNBAQOFJWCA-UAKXSSHOSA-N
正在寻找类似产品? 访问 产品对比指南
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
个人防护装备
Eyeshields, Gloves
其他客户在看
Angelica M Bello et al.
Journal of medicinal chemistry, 52(6), 1648-1658 (2009-03-06)
A series of 6-substituted and 5-fluoro-6-substituted uridine derivatives were synthesized and evaluated for their potential as anticancer agents. The designed molecules were synthesized from either fully protected uridine or the corresponding 5-fluorouridine derivatives. The mononucleotide derivatives were used for enzyme
Inhibition of RNA synthesis by 5-fluorouridine accounts for its cyto toxicity on colorectal cancer cells in vitro
Subbarayan PR, et al.
Cancer research, 65(9) (2005)
P V Sahasrabudhe et al.
Nucleic acids research, 23(19), 3916-3921 (1995-10-11)
The effects of 5-fluorouridine (FUrd) and 5-fluorodeoxyuridine (FdUrd) substitution on the stabilities of duplex RNA and DNA have been studied to determine how FUrd substitution in nucleic acids may alter the efficiency of biochemical processes that require complementary base pairing
Nikolaos Tsesmetzis et al.
Cancers, 10(7) (2018-07-26)
Antimetabolites, in particular nucleobase and nucleoside analogues, are cytotoxic drugs that, starting from the small field of paediatric oncology, in combination with other chemotherapeutics, have revolutionised clinical oncology and transformed cancer into a curable disease. However, even though combination chemotherapy
Mingjie Chen et al.
The Plant cell, 23(8), 2991-3006 (2011-08-11)
Nucleotides are synthesized from de novo and salvage pathways. To characterize the uridine salvage pathway, two genes, UKL1 and UKL2, that tentatively encode uridine kinase (UK) and uracil phosphoribosyltransferase (UPRT) bifunctional enzymes were studied in Arabidopsis thaliana. T-DNA insertions in
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门