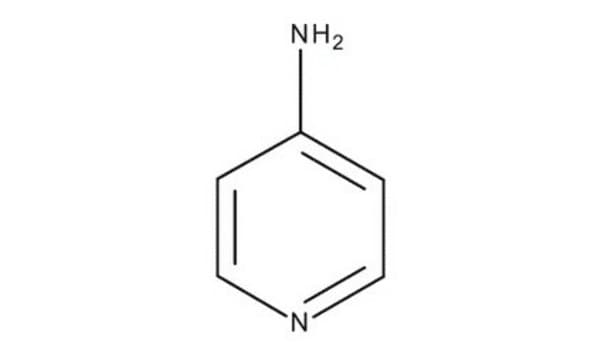
价格与库存信息目前不能提供
推荐产品
化驗
99%
形狀
flakes
bp
248 °C (lit.)
mp
60-63 °C (lit.)
SMILES 字串
Nc1cccnc1
InChI
1S/C5H6N2/c6-5-2-1-3-7-4-5/h1-4H,6H2
InChI 密鑰
CUYKNJBYIJFRCU-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
訊號詞
Danger
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT RE 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
190.4 °F - closed cup
閃點(°C)
88 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Chao Fang et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 71(4), 1588-1593 (2008-07-22)
Our algorithm [B. Tian, G. Wu, G. Liu, J. Chem. Phys. 87 (1987) 7300] is introduced to obtain the temporal bond polarizabilities of 2- and 3-aminopyridine from their Raman intensities, which supply fruitful electronic information of the nonresonant Raman excited
Elizabeta Sauer et al.
Antimicrobial agents and chemotherapy, 48(12), 4532-4541 (2004-11-25)
The utilization pathway for the uptake of NAD and nicotinamide riboside was previously characterized for Haemophilus influenzae. We now report on the cellular location, topology, and substrate specificity of PnuC. pnuC of H. influenzae is only distantly related to pnuC
Effects of chronic, intrauterine organic and inorganic mercury intoxication on the epileptogenicity of developing rat.
B Barna et al.
Central European journal of public health, 8 Suppl, 73-75 (2000-08-16)
B Barna et al.
Acta biologica Hungarica, 50(1-3), 257-267 (1999-11-26)
The effect of GYKI 52466, a selective, non-competitive antagonist of the AMPA glutamate receptor subtype was investigated on the development, expression and propagation of 3-aminopyridine-induced cortical ictal activity, both in the primary and secondary focus. In one group of animals
E O'Hearn et al.
The Journal of neuroscience : the official journal of the Society for Neuroscience, 17(22), 8828-8841 (1997-11-14)
Ibogaine, an indole alkaloid that causes hallucinations, tremor, and ataxia, produces cerebellar neurotoxicity in rats, manifested by degeneration of Purkinje cells aligned in narrow parasagittal bands that are coextensive with activated glial cells. Harmaline, a closely related alkaloid that excites
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门