MSQC9
SILu™MAb Infliximab Stable-Isotope Labeled Monoclonal Antibody
recombinant, expressed in CHO cells
Synonym(e):
Mass spectrometry standard, Infliximab, SIL Infliximab, Stable isotope labelled Infliximab
About This Item
Empfohlene Produkte
Rekombinant
expressed in CHO cells
Qualitätsniveau
Antikörper-Produkttyp
primary antibodies
Assay
≥90% (SDS-PAGE)
Verpackung
vial of 100 μg
Versandbedingung
wet ice
Lagertemp.
−20°C
Allgemeine Beschreibung
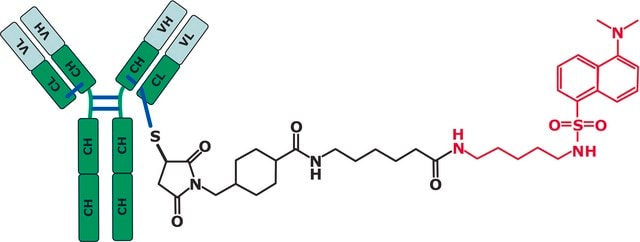
Infliximab has been approved for the treatment of Crohn′s disease, ulcerative colitis, psoriasis, psoriatic arthritis, ankylosing spondylitis, and rheumatoid arthritis. SILu™MAb Infliximab Stable-Isotope Labeled Monoclonal Antibody is for R&D use only. Not for drug, household, or other uses.
Anwendung
Physikalische Form
Angaben zur Herstellung
Rekonstituierung
- Briefly centrifuge the vial at ~10,000 × g to collect the product at the bottom of the vial.
- Add 500 μL of ultrapure water containing 0.1% formic acid to the vial.
- Mix the contents by gently inverting the vial a minimum of 5 times.
- Allow the vial to stand at room temperature for at least 15 minutes and repeat mixing by inversion.
Hinweis zur Analyse
MRM settings provided (xls)
Rechtliche Hinweise
Sie haben nicht das passende Produkt gefunden?
Probieren Sie unser Produkt-Auswahlhilfe. aus.
Lagerklassenschlüssel
13 - Non Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.