Alle Fotos(1)
Wichtige Dokumente
275778
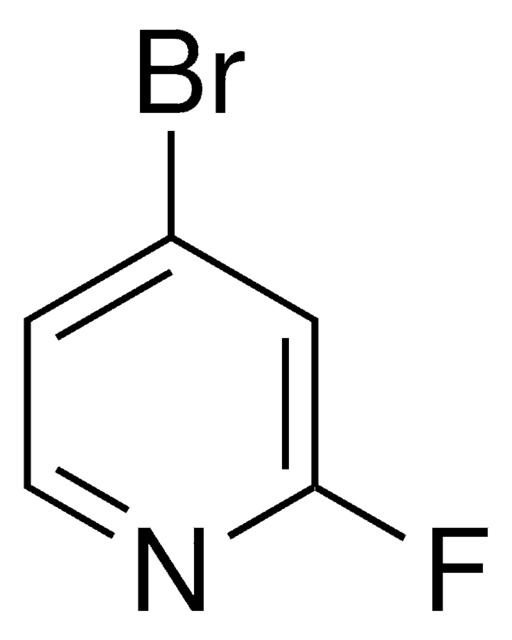
Imidazo[1,2-a]pyridin
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C7H6N2
CAS-Nummer:
Molekulargewicht:
118.14
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
99%
Form
liquid
Brechungsindex
n20/D 1.626 (lit.)
bp
103 °C/1 mmHg (lit.)
Dichte
1.165 g/mL at 25 °C (lit.)
SMILES String
c1ccn2ccnc2c1
InChI
1S/C7H6N2/c1-2-5-9-6-4-8-7(9)3-1/h1-6H
InChIKey
UTCSSFWDNNEEBH-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
In vivo anti-trypanosomal activity of imidazo[1,2-a]pyridiness in the STIB900 mouse model has been investigated.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
10 - Combustible liquids
WGK
WGK 3
Flammpunkt (°F)
235.4 °F - closed cup
Flammpunkt (°C)
113 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Martina Hieke et al.
Bioorganic & medicinal chemistry letters, 22(5), 1969-1975 (2012-02-14)
A novel class of 5-lipoxygenase (5-LO) inhibitors characterized by a central imidazo[1,2-a]pyridine scaffold, a cyclohexyl moiety and an aromatic system, is presented. This scaffold was identified in a virtual screening study and exhibits promising inhibitory potential on the 5-LO. Here
Joanna M Wisniewska et al.
Biochemical pharmacology, 83(2), 228-240 (2011-10-27)
5-Lipoxygenase (5-LO) is a crucial enzyme of the arachidonic acid (AA) cascade and catalyzes the formation of bioactive leukotrienes (LTs) which are involved in inflammatory diseases and allergic reactions. The pathophysiological effects of LTs are considered to be prevented by
Enza Palazzo et al.
Neuropharmacology, 58(3), 660-667 (2009-12-01)
The 6-methoxy-2-phenylimidazo[1,2-b]pyridazine-3-carboxylic acid, DM2, exerts anti-absence activity and blocks Cav3.1 channel, a T-type voltage-dependent Ca(2+) channel subtype, in vitro. The current study investigated the effect of intra-ventrolateral periaqueductal grey (VLPAG) administration of DM2 on formalin-induced nocifensive responses in rats. In
Sophie Marhadour et al.
European journal of medicinal chemistry, 58, 543-556 (2012-11-21)
A novel series of 2,3-diarylimidazo[1,2-a]pyridines was synthesized and evaluated for their antileishmanial activities. Four derivatives exhibited good activity against the promastigote and intracellular amastigote stages of Leishmania major, coupled with a low cytotoxicity against the HeLa human cell line. The
Kyle A Emmitte et al.
Bioorganic & medicinal chemistry letters, 19(3), 1004-1008 (2008-12-23)
The optimization of imidazo[1,2-a]pyridine inhibitors as potent and selective inhibitors of IGF-1R is presented. Further optimization of oral exposure in mice is also discussed. Detailed selectivity, in vitro activity, and in vivo PK profiles of an optimized compound is also
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.![2-phenylimidazo[1,2-a]pyridine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/281/247/6c2550a0-2f0c-4866-83d8-3c1fb039e165/640/6c2550a0-2f0c-4866-83d8-3c1fb039e165.png)
![Imidazo[1,2-a]pyrazin 97%](/deepweb/assets/sigmaaldrich/product/structures/370/804/1712d71f-52fb-4758-9a22-85b6c96cd4e8/640/1712d71f-52fb-4758-9a22-85b6c96cd4e8.png)
![Imidazo[1,2-a]pyrimidine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/187/001/4862c14e-bec7-4475-85a5-f178e48ff60f/640/4862c14e-bec7-4475-85a5-f178e48ff60f.png)