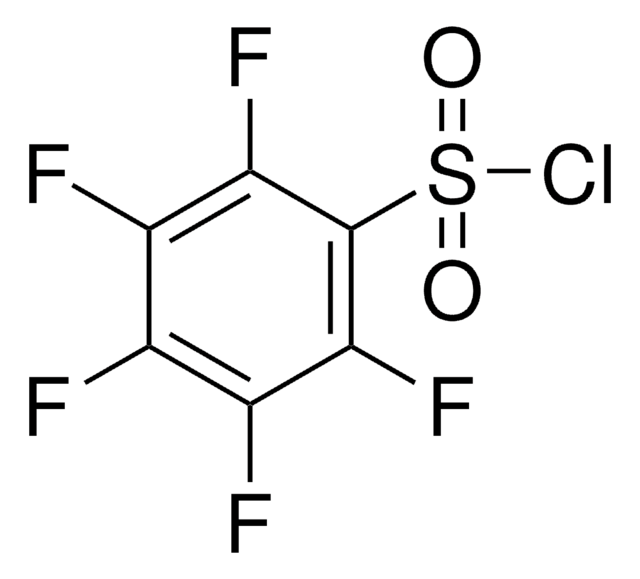
F6206
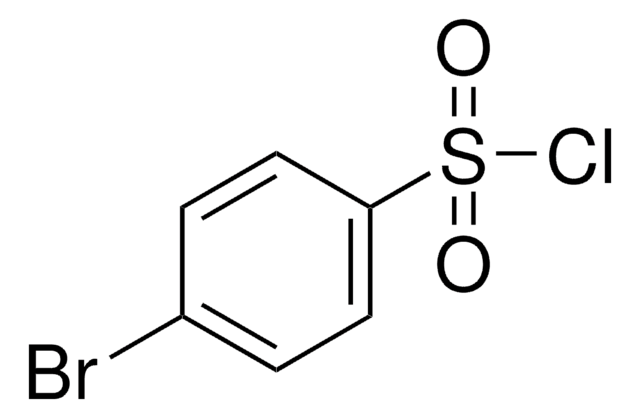
4-Fluorobenzenesulfonyl chloride
98%
Synonym(s):
4-Fluorobenzene-1-sulfonyl chloride, p-Fluorobenzenesulfonyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
FC6H4SO2Cl
CAS Number:
Molecular Weight:
194.61
Beilstein:
2091633
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
bp
95-96 °C/2 mmHg (lit.)
mp
29-31 °C (lit.)
SMILES string
Fc1ccc(cc1)S(Cl)(=O)=O
InChI
1S/C6H4ClFO2S/c7-11(9,10)6-3-1-5(8)2-4-6/h1-4H
InChI key
BFXHJFKKRGVUMU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Y A Chang et al.
Bioconjugate chemistry, 3(2), 200-202 (1992-03-01)
4-Fluorobenzenesulfonyl chloride (fosyl chloride), due to the strong electron-withdrawing property of its fluoride atom, is found to be an excellent activating agent for the covalent attachment of biologicals to a variety of solid supports (e.g. functionalized polystyrene microspheres, Sepharose beads
T H Liao et al.
Analytical biochemistry, 148(2), 365-375 (1985-08-01)
The reagent p-fluorobenzenesulfonyl chloride modifies the protein side chains of tyrosine, lysine, and histidine and the alpha-NH2 group. The p-fluorobenzenesulfonyl (Fbs-) group, identified by the 19F nuclear magnetic resonance signal, exhibits a different 19F chemical shift for each functional group
Umaprasana Ojha et al.
Langmuir : the ACS journal of surfaces and colloids, 25(11), 6319-6327 (2009-04-02)
Peptide surface modification of poly[(methyl methacrylate-co-hydroxyethyl methacrylate)-b-isobutylene-b-(methyl methacrylate-co-hydroxyethyl methacrylate)] P(MMA-co-HEMA)-b-PIB-b-P(MMA-co-HEMA) triblock copolymers with different HEMA/MMA ratios has been accomplished using an efficient synthetic procedure. The triblock copolymers were reacted with 4-fluorobenzenesulfonyl chloride (fosyl chloride) in pyridine to obtain the activated
Ning Li et al.
European journal of medicinal chemistry, 155, 531-544 (2018-06-18)
Ten novel symmetric 3,5-bis(arylidene)-4-piperidone derivatives (BAPs, 1-10) and fourteen dissymmetric BAPs (11-24) were synthesized and evaluated the cytotoxicity. All of the compounds have been screened for their anti-inflammatory activity characterized by evaluating their inhibitory effects on LPS-induced IL-6, TNF-α secretion.
Abdelaziz Houmam et al.
Physical chemistry chemical physics : PCCP, 14(1), 113-124 (2011-09-15)
Important aspects of the electrochemical reduction of a series of substituted arene sulfonyl chlorides are investigated. An interesting autocatalytic mechanism is encountered where the starting material is reduced both at the electrode and through homogeneous electron transfer from the resulting
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service