566357
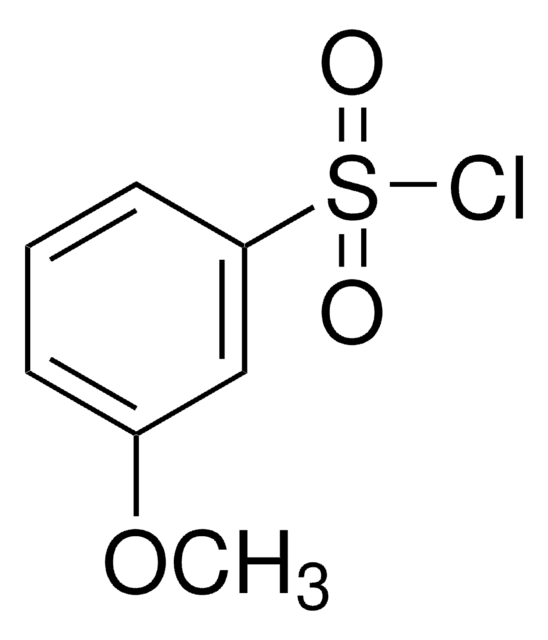
m-Toluenesulfonyl chloride
97%
Synonym(s):
3-Methylbenzenesulfonyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3C6H4SO2Cl
CAS Number:
Molecular Weight:
190.65
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
refractive index
n20/D 1.5490 (lit.)
bp
252-253 °C (lit.)
density
1.314 g/mL at 25 °C (lit.)
SMILES string
Cc1cccc(c1)S(Cl)(=O)=O
InChI
1S/C7H7ClO2S/c1-6-3-2-4-7(5-6)11(8,9)10/h2-5H,1H3
InChI key
KFPMLWUKHQMEBU-UHFFFAOYSA-N
Related Categories
General description
m-Toluenesulfonyl chloride can be synthesized via chlorination of m-thiocresol in glacial acetic acid.
Application
m-Toluenesulfonyl chloride may be used in the synthesis of mesityl-m-tolyl sulfone via reaction with mesitylene.
Reactant involved in they synthesis of biologically active molecules including:
- Small molecule ligands for the Stat3 SH2 domain
- Isoindoline-5-propenehydroxamates for use as histone deactylase inhibitors
- Phenyl-pyrazolylamine-based derivatives as FLT3 kinase inhibitors
- Aryldisulfonamides with antibacterial activity
- Quinazoline analogs for the treatment of Gaucher disease
- 17β-HSD2 inhibitors for the treatment of osteoporosis
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
225.0 °F - closed cup
Flash Point(C)
107.2 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Rearrangements of sulfones to sulfinic acids via carbanion intermediates.
Truce WE and Brand WW
The Journal of Organic Chemistry, 35(6), 1828-1833 (1970)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service