모든 사진(1)
크기 선택
보기 변경
5 G
₩120,785
About This Item
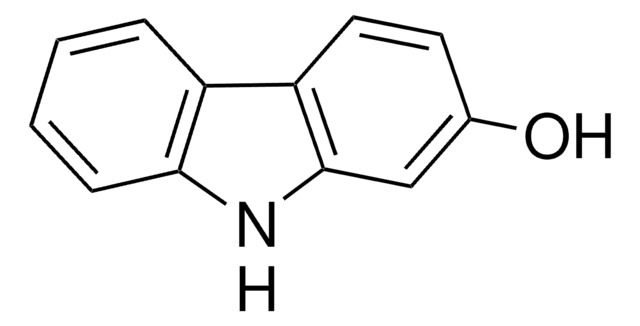
실험식(Hill 표기법):
C12H9NO
CAS Number:
Molecular Weight:
183.21
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
95%
mp
169-173 °C (lit.)
SMILES string
Oc1cccc2[nH]c3ccccc3c12
InChI
1S/C12H9NO/c14-11-7-3-6-10-12(11)8-4-1-2-5-9(8)13-10/h1-7,13-14H
InChI key
UEOHATPGKDSULR-UHFFFAOYSA-N
일반 설명
4-Hydroxycarbazole can be obtained from 1,2,3,4-tetrahydro-4-oxocarbazole via dehydrogenation with freshly prepared Raney nickel.[1]
애플리케이션
4-Hydroxycarbazole may be used in the synthesis of the following:[2]
It participates as an electron donor for the preparation of nonlinear optical (NLO) chromophores.[3]
- 4-(2-bromoethoxy)-9H-carbazole
- 4-(3-bromopropoxy)-9H-carbazole
- 4-(4-bromobutoxy)-9H-carbazole
- 4-(5-bromopentyloxy)-9H-carbazole
- 4-(6-bromohexyloxy)-9H-carbazole
It participates as an electron donor for the preparation of nonlinear optical (NLO) chromophores.[3]
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
Kristan H Cleveland et al.
PloS one, 14(5), e0217038-e0217038 (2019-05-21)
Carvedilol is reported to prevent cancers in humans and animal models. However, a molecular mechanism has yet to be established, and the extent to which other β-blockers are chemopreventive remains relatively unknown. A comparative pharmacological approach was utilized with the
Pramod V Chavan et al.
Bioorganic chemistry, 85, 475-486 (2019-02-19)
A series of spirochromenocarbazole tethered 1,2,3-triazoles were synthesized via click chemistry based one-pot, five component reaction between N-propargyl isatins, malononitrile, 4-hydroxycarbazole, aralkyl halides and sodium azide using cellulose supported CuI nanoparticles (Cell-CuI NPs) as the heterogeneous catalyst. Antiproliferative activity of
Synthesis and characterization of novel electro-optic chromophores based on 4-hydroxycarbazole.
Zhou T, et al.
Materials Letters, 97, 117-120 (2013)
Kevin M Huang et al.
Cancer prevention research (Philadelphia, Pa.), 10(10), 598-606 (2017-09-16)
In previous studies, the β-blocker carvedilol inhibited EGF-induced epidermal cell transformation and chemical carcinogen-induced mouse skin hyperplasia. As exposure to ultraviolet (UV) radiation leads to skin cancer, the present study examined whether carvedilol can prevent UV-induced carcinogenesis. Carvedilol absorbs UV
K Zawadzka et al.
Environmental science and pollution research international, 22(24), 19658-19666 (2015-08-16)
Nitrogen heterocyclic compounds, especially carbazole, quinolone, and pyridine are common types of environmental pollutants. Carbazole has a toxic influence on living organisms, and the knowledge of its persistence and bioconversion in ecosystems is still not complete. There is an increasing
활성 필터
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.