추천 제품
분석
99%
형태
powder with small lumps
광학 활성
[α]25/D −26.4°, c = 1 in H2O
색상
white
mp
272 °C (lit.)
응용 분야
peptide synthesis
저장 온도
2-8°C
SMILES string
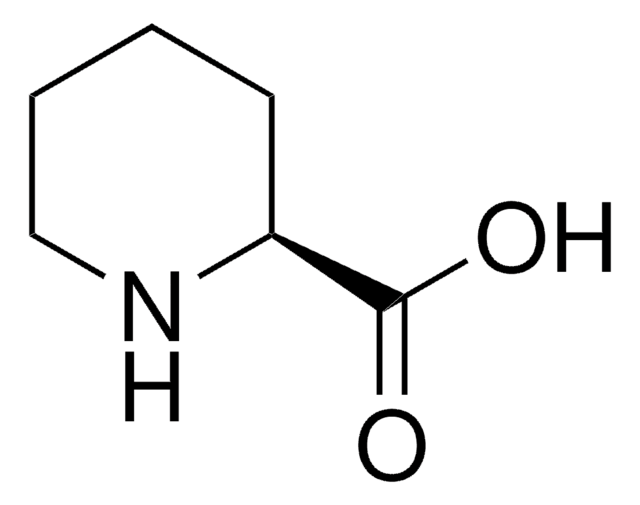
OC(=O)[C@@H]1CCCCN1
InChI
1S/C6H11NO2/c8-6(9)5-3-1-2-4-7-5/h5,7H,1-4H2,(H,8,9)/t5-/m0/s1
InChI key
HXEACLLIILLPRG-YFKPBYRVSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Occurs in seeds, malt, edible mushrooms, fruits, etc.
교체됨
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
M R Baumgartner et al.
Annals of neurology, 47(1), 109-113 (2000-01-13)
We describe an 18-year-old patient with psychomotor retardation and abnormally short metatarsi and metacarpals but no other signs of classic Refsum disease. Molecular analysis of the phytanoyl-coenzyme A hydroxylase gene revealed a homozygous deletion causing a frameshift. Surprisingly, L-pipecolic acid
D W Armstrong et al.
Journal of pharmaceutical and biomedical analysis, 11(10), 881-886 (1993-10-01)
Recently it was found that normal adults excrete pipecolic acid primarily as the D-enantiomer even though it is present in the blood stream mainly as the L-enantiomer (i.e. > 98% L). This study of pipecolic acid stereochemistry was extended to
S J Mihalik et al.
Pediatric research, 25(5), 548-552 (1989-05-01)
L-Pipecolic acid, a cyclic imino acid produced during the degradation of lysine, accumulates in body fluids of infants with the generalized peroxisomal disorders, including Zellweger syndrome, neonatal adrenoleukodystrophy, and infantile Refsum disease. Peroxisome-enriched fractions from normal human liver oxidized L-[3H]pipecolic
Hai-Chao Xu et al.
Journal of the American Chemical Society, 132(8), 2839-2844 (2010-02-06)
Anodic olefin coupling reactions using a tosylamine trapping group have been studied. The cyclizations are favored by the use of a less-polar radical cation and more basic reaction conditions. The most significant factor for obtaining good yields of cyclic product
Eduard A Struys et al.
FEBS letters, 584(1), 181-186 (2009-11-26)
The mammalian degradation of lysine is believed to proceed via two distinct routes, the saccharopine and the pipecolic acid routes, that ultimately converge at the level of alpha-aminoadipic semialdehyde (alpha-AASA). alpha-AASA dehydrogenase-deficient fibroblasts were grown in cell culture medium supplemented
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.